Abstract:
Background: General practitioners (GP) play a crucial role in identifying the signs and symptoms of many febrile illnesses, including dengue and also provide care and referral to hospital for severe cases. This study aimed to identify the prevalence of Dengue infection among children with acute febrile illness in GP clinics at Peri-urban settings, Yangon, to determine the clinical and hematological predictors of acute dengue infection and to verify the diagnostic accuracy of WHO 2009 criteria for probable dengue.
Methodology: It was a cross-sectional analytical study in 408 children (2-15 years old) with 1- 7 days of fever visiting GP clinics during June 2017 to August 2018. Clinical presentations of the patients were asked and examined by the GP and recorded in the proforma. Probable dengue was defined in accordance to 2009 WHO criteria. Acute dengue infection was determined if either NS1 or Ig M (or both) is positive by using SD (Bioline) Rapid Diagnostic Test for dengue.
Results: The prevalence of acute dengue infection among children with acute febrile infection was 22.5% (92/408). Aches and pain, leukopenia and increase in haematocrit concurrent with platelet counts were independently predictors of acute dengue infection. The diagnostic accuracy of 2009 WHO criteria for probable dengue was as follow; sensitivity 73.9% (63.7-85.5), specificity 57% (51.3- 62.5), positive predictive value 29.6 (33.3-37.3) and negative predictive value 83.9% (88.2-91.5).
Conclusions: GP should have high suspicion of dengue infection if children present with aches and pain and have leukopenia and increase in hematocrit concurrent with decrease in platelet count.
Key-words: dengue fever, children, predictors, WHO 2009 guideline
Introduction
Dengue is the fastest growing mosquito-borne disease, and is also one of the leading causes of serious illness and death among children1. According to World Health Organization, approximately 390 million people were estimated to have dengue virus infection, of which, 96 million showed clinical manifestations1. It can also lead to over 5900 deaths in South East Asian countries annually2.
In Myanmar, the first case of dengue was found in Yangon in 1965, and the country had experienced dengue outbreaks since 19703. The disease is endemic every two or three years, and the number of cases became highest in 2007, 2011, 2013 and 2015 4. The reported case fatality rate of dengue in Myanmar was slightly above 1% 5. From January to 1st December in 2016, Yangon, the largest city of Myanmar, had the highest number of dengue cases (1177 reported patients) among all divisions over the country and had 6 mortality cases 4.
Although dengue hemorrhagic fever cases are usually highest in rainy season, the incidence occurs throughout the year in Myanmar. The citizens in Yangon are used to visit a general practitioner (GP) first in case of illness. GPs have a vital and active role to play in identifying the signs and symptoms of many febrile illnesses including dengue and also to provide care and referral to hospital in severe cases. The clinical signs and symptoms of Dengue are usually mild and mostly present with undifferentiated febrile illness. Early identification of dengue infection could help medical practitioners to give early supportive therapies6, prevent the use of potentially harmful drugs 7, and encourage assessment of prognosis and the use of the treatment guidelines 8.
In 2009, World Health Organization proposed new diagnostic criteria for probable dengue infection with variable sensitivity and specificity in different populations 9, 10. In Myanmar, although some studies have identified signs and symptoms associated with confirmed dengue cases, the evaluation of multiple clinical criteria to predict dengue infection has yet been very limited, especially in the population visiting GP clinics, in early non-critical stage of illness. Following clinical criteria to detect dengue should be a practice in a developing country like Myanmar because of the cost and limited availability of the rapid diagnostic tests in many parts of the country. However, in a recent study on point of care testing for diagnosis of dengue in 72 GPs in Yangon, only 27.9% of them apply WHO clinical criteria in diagnosis of dengue 11.
This study aimed to identify the prevalence of Dengue infection among children with acute febrile illness in GP clinics in Periurban settings of Yangon, to determine the clinical and haematological predictors of dengue infection from the parameters proposed in WHO 2009 guideline as probable dengue and to verify the diagnostic accuracy of WHO 2009 criteria for probable dengue.
Materials and Methods
A cross-sectional, analytical study was conducted on 408 children visiting some GP clinics in periurban settings of Yangon from June 2017 to August, 2018. Consecutive sampling was done in all children (2-15 years old) who visited to the assigned GP clinics with acute 1-7 days of fever. Patients with known immunocompromised status (e.g. Taking steroids, chemotherapy, malignancy, HIV, chronic organic diseases) were excluded from the study. Informed consent was taken from caregivers and informed assent was taken from children older than 10 years old. Patients were asked and examined the clinical presentation according to proforma by the General Practitioner. Blood sample were taken from the participants by a trained research assistant.
Approximately 3 ml of blood was collected (1 ml of clotted blood in plain tube for Dengue RDT and 2 ml of un-coagulated blood in K3ethylenediaminetetraacetic acid (EDTA) tubes for complete blood count). Rapid diagnostic test for Dengue was tested by SD Bioline Dengue RDT test kit at the GP clinic and blood for complete picture will be analyzed by coulter counter machine at the assigned laboratory within 24 hours after taking blood samples. Baseline information, clinical presentations and laboratory results was recorded by using the proforma. Probable dengue was defined in accordance to 2009 WHO criteria (fever and 2 of the following: nausea/vomiting, rash, aches and pain, tourniquet test positive, leukopenia and any warning signs such as abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation, mucosal bleed, lethargy, restlessness, liver enlargement >2 cm, increase in haematocrit concurrent with rapid decrease in platelet count) 5. Leukopenia was defined if WBC count is lower than 5 x 109/mm, increase in heatocrit was defined if haematocrit value was ≥ 44%, and decrease in platelet count was defined if platelet count was ≤100x 103/L. Acute dengue infection was confirmed in all participants by using Rapid Diagnostic Test for dengue. Acute dengue infection was determined if either NS1 or Ig M (or both) is positive.
Statistical analysis
The data was subjected to statistical analysis using the STATA 12 software package and evaluated using descriptive statistics (mean and standard deviation, median, frequency and percentages as appropriate). Comparisons between clinical and laboratory parameters and presence or absence of Dengue infection by RDT results will be made using Chi-squared or Fisher exact tests. Predictors of dengue infection was analyzed from the clinical and hematological parameters proposed by WHO 2009 guideline as probable dengue by putting data into stepwise logistic regression analysis. Diagnostic accuracy of probable dengue criteria in WHO 2009 guideline and set of predictors from multivariable logistic regression was described by sensitivity, specificity and positive and negative predictive values.
Ethical Review
This study was approved by Ethics Review Committee, Department of Medical Research.
Results
The study comprised of 408 patients with acute febrile illness (mean age 7.6 ± 3.7 year). Males accounted for 57.4% (234/408) and females accounted for 42.6%(174/408). The prevalence of acute dengue infection was 22.5% (92/408), in which NS1 Ag was positive in 59 (14.5%) and IgM Ab is posititve in 45 (11%). IgG Ab was positive in 143 (35%).
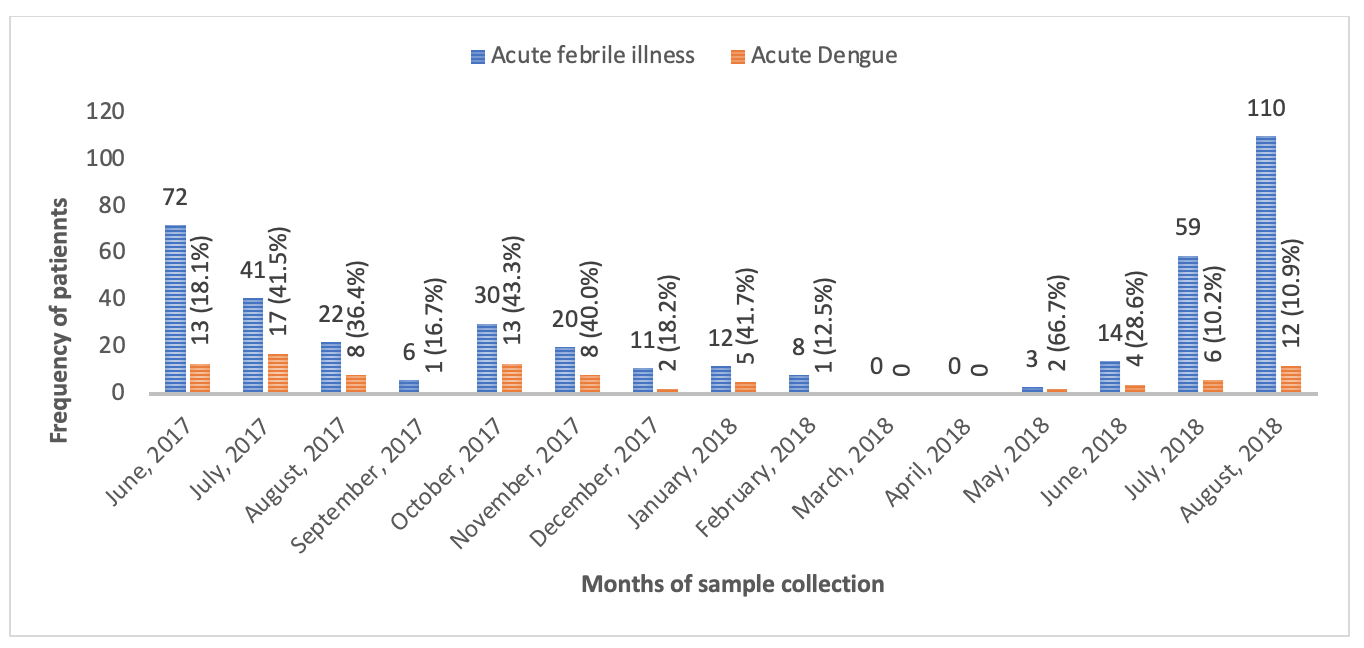
Fig 1. Distribution of acute febrile illness and proportion of acute dengue cases within 15 months period
Apart from March and April, 2018 in which no cases were collected for the study, acute dengue infection was seen in all months during sample collection (Figure 1).
Table 1-Comparison of baseline characteristics in febrile children with and without acute dengue infection
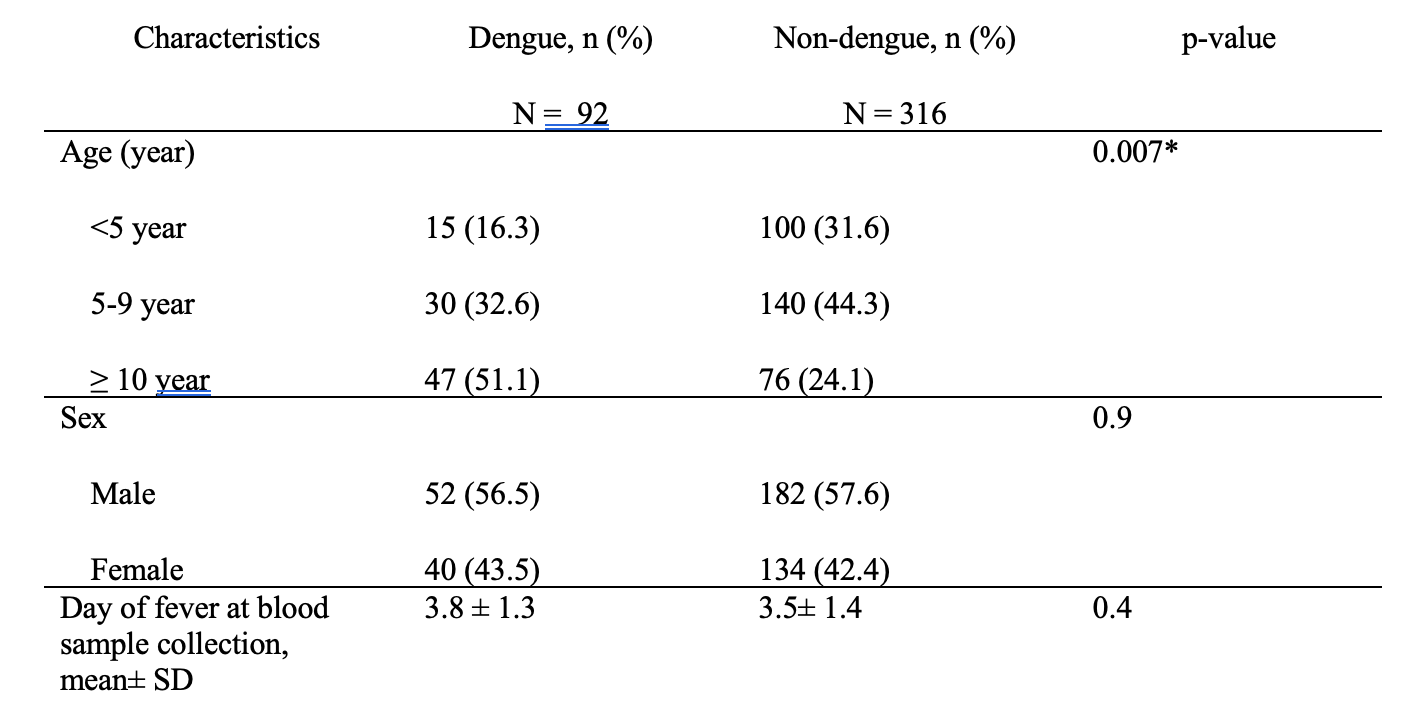
In this study, the highest proportion of dengue cases was seen in ≥ 10 year age group (51.1%) followed by 5-9 years age group (32.6%) and <5 years age group (16.3%). The difference in proportion of dengue infection between age groups was statistically significant. There was no difference in finding dengue infection among sexes and among days of fever at blood sample collection (Table 1).
Table 2-Comparison of clinical and haematological parameters of probable dengue in febrile children with and without acute dengue infection
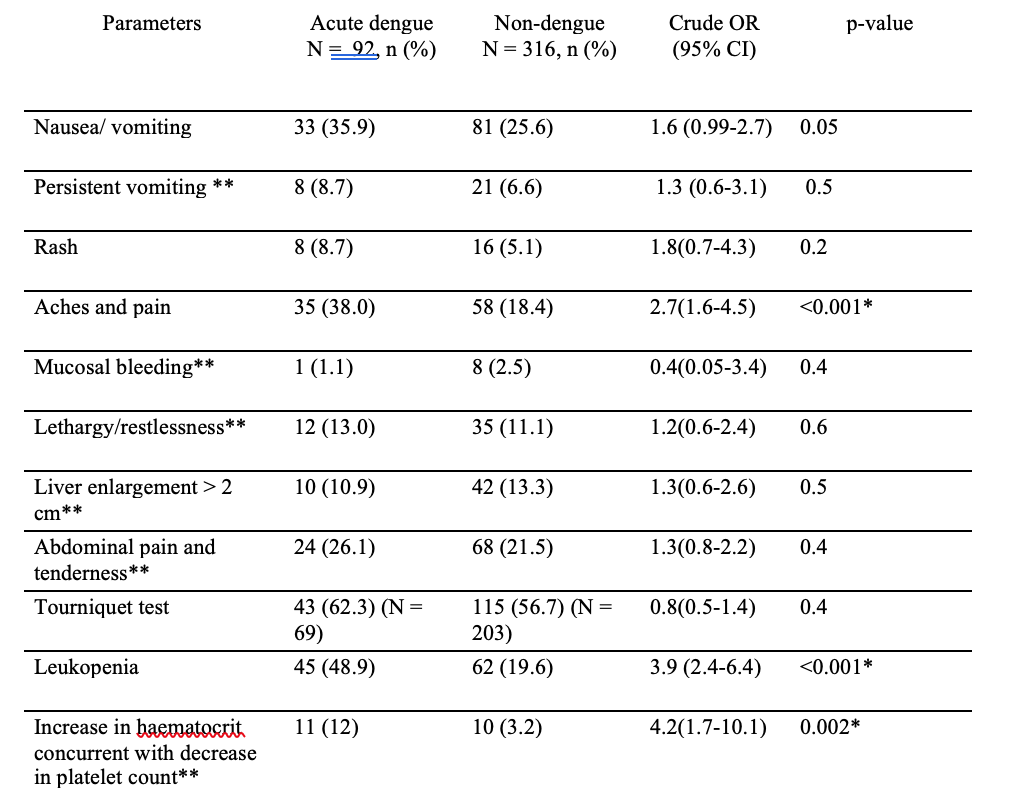
*statistically significant
**warning signs
Dengue infection was significantly higer in patients having aches and pain, leukopenia and increase in hematocrit concurrent with decrease in platelet count (Table 2).
Table 3-Parameters independently associated with acute dengue infection

*statistically significant
**warning signs
On multivariable logistic regression, the three parameters (aches and pain, leukopenia and increase in hematocrit concurrent with decrease in platelet count) were found to be independently associated with acute dengue infection (Table 3).
Table 4-Diagnostic accuracy of probable dengue in WHO 2009 criteria in the study population

WHO 2009 criteria for probable dengue had moderate sensitivity (73.9%) and negative predictive value (88.2%) but low specificity (57%) and positive predictive value (33.3%) to diagnose acute dengue infection comparing with the laboratory confirmation by Dengue Duo RDT.
Discussion
In the study population, acute dengue infection can be seen in all seasons in GP clinics in Myanmar and the number of collected cases were highest in the months of rainy season. Pwint Mon Oo et al also described that the majority of dengue cases occurred in the wet season from May to September13. The findings were concurrent with the knowledge of seasonal transmission in which high temperatures, relative humidity, heavy rainfall in the wet season, and poor drainage can cause the multiplication of the main vector, Aedes aegypti, in urban environment, and also favor the propagation of dengue viruses 14, 15. The finding of dengue infection throughout the year in the study population pointed out that clinicians should aware of the probability of dengue infection not only in rainy season but also in the whole year in Yangon.
In this study, the proportion of acute dengue infection varied by age group with approximately a 2-found difference each between children <5 years old, 5-9-year-old and ≥ 10 year (16.3% vs 32.6% vs 51.1%). The increasing trend of dengue infection related to older age group in this study was concordant with the findings in a Puerto Rico study, in which dengue incidence varied by age group with a nearly a 10-fold difference between participants <5 years old and 10±19 years old (3% versus 27%). The higher chance to get secondary dengue infection in older children may lead to seek Medicare at GPs than younger children, who could more likely to have milder primary infection, may be the reason of this finding 16.
This study found that some probable dengue parameters presenting to GPs were useful to predict acute dengue infection in Myanmar children. Having aches and pain was the only clinical parameter which can predict acute dengue infection in children in this study. Joint pain was a predicting factors of dengue vs other acute febrile illness in adults at Sri-Lannka 17. This study also evaluated the ability of hematological parameters (leukopenia and increase in hematocrit concurrent with decrease in platelet count) to discriminate dengue from other acute febrile illness. The relevance of leukopenia as a discriminant feature of dengue infection has been previously documented, although the cutoff values may vary 18, 19, 20. Leukopenia was also found to a predictive factor of dengue illness in Brazil 21.A systematic review also reported that low platelet counts and decreases in WBC were independently associated with the presence of dengue 22. In 2018, National guideline for clinical management of dengue was published by Ministry of Health in Sports, Myanmar. The guideline describes leukopenia as a good predictor of dengue. It also recommends to test complete blood count not only in patients with warning signs and circulatory failure but also in all patients at first visit and patients with fever > 3 days 23. For a dengue endemic area like Myanmar, early case identification of dengue is important to prevent the mortality from severe cases. However, rapid diagnostic tests are not recommended as compulsory test in the National guideline. These tests are not very economic for every people in resource-poor countries; therefore, it will reduce the cost of laboratory diagnostic test if the GPs can clinically determine who should be sent for the dengue confirmatory test and who should be followed up with great care in daily clinical practice depending on the simple clinical and basic heamtological parameters from Complete Blood Count (CBC). According to the findings in this study, GPs should have high suspicion of and should confirm acute dengue infection if the children presents with aches and pain or have leukopenia or increase in hematocrit and decrease in platelet count at CBC.
WHO 2009 criteria have variable sensitivity and specificity in children with dengue infection. To our knowledge, this is the first evaluation of the performance of the 2009 WHO dengue classification criteria at identifying acute dengue infection among children visiting GP clinics with acute febrile illness in Yangon, Myanmar. Regarding diagnostic accuracy of WHO 2009 criteria in children with acute dengue illness, the sensitivity (73.9%) and specificity (57%) of this study is comparable to those in Singaporean adults with acute febrile illness (sensitivity 79.7% and sensitivity 57%) 24. In Sri-Lankan children, the WHO 2009 criteria showed slightly lower sensitivity (69.9%) but higher specificity (75.1%) than our study 17. A Brazilian study in adults reported the higher sensitivity 87.1 % and lower specificity 24.3 % for WHO 2009 criteria 21 and a Singaporean study in adults also reported similar sensitivity (87.1%) and specificity (26%) 25. The difference in diagnostic accuracy of WHO 2009 probable dengue criteria may be due to the difference in prevalences of dengue and other febrile illnesses in the region which can affect sensitivity and specificity of clinical parameters. The lower diagnostic accuracies of WHO 2009 criteria in this study indicates that it is not the best to screen acute dengue infection in GPs using WHO 2009 criteria of probable dengue. Compared to the probable dengue criteria in 2009 WHO classification, dengue case definition in 1997 WHO classification had higher sensitivity in a study26 and was suggested to be useful in ruling out dengue. Case definition of dengue in recent National guideline in Myanmar was based on 1997 WHO classification. Additional prospective studies should be done in Myanmar patients with acute febrile illness in different population at different health care settings for the predictive capacity of specific clinical features in the new National guideline, because an easily applicable clinical algorithm could have a favorable impact on the economies of dengue-endemic developing countries.
The main limitation of this study was that acute dengue infection was confirmed in acute samples only by using dengue duo RDT. The gold standard method for dengue diagnosis is to test paired serum samples in both acute and convalescent period for IgM and IgG using ELISA. Both NS1 and IgM tests were reported to have lower sensitivities in secondary infections. As dengue is endemic in Myanmar, without testing convalescent samples, it is possible that some secondary dengue patients could be classified as non-dengue. This may lead to underestimation of the accuracy of the analyzed predictors because sensitivity and specificity of clinical parameters depend on the prevalence of the disease. This study was conducted in GPs where there are more mild cases compared to hospital setting; therefore, the findings may not represent the population in hospitals to where the suspected dengue cases were referred leading higher prevalence of both severe and non-severe dengue.
Conclusion
In dengue endemic areas like Yangon, dengue fever should be considered in all patient presenting with an acute undifferentiated febrile illness during any season. GPs should have high suspicion of dengue infection if children present with aches and pain and have leukopenia and increase in hematocrit concurrent with decrease in platelet count. As the diagnostic accuracy of probable dengue criteria in WHO 2009 classification was not very high in this study, applicability of more clinical parameters in diagnosis of dengue should be studied in different Myanmar populations.
References
1. Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, et al. (2013).The global distribution and burden of dengue. Nature.496(7446):504-7.
2. Shepard DS, Undurraga EA, Halasa YA. (2013) Economic and disease burden of dengue in Southeast Asia. PLoS neglected tropical diseases.7(2):e2055.
3. Thein, S., (1991). Dengue haemorrhagic fever in Myanmar. DMR Bull. 5, 1–14.
4. Myanmar likely to experience dengue epidemic next year Eleven (online). Available at: http://www.elevenmyanmar.com/local/6936
5. WHO. Dengue: Guidelines for diagnosis, treatment, prevention and control 2009.
6. Harris E, Perez L, Phares CR, Perez Mde L, Idiaquez W, Rocha J, Cuadra R, Hernandez E, Campos LA, Gonzales A, et al: (2003); Fluid intake and decreased risk for hospitalization for dengue fever, Nicaragua. Emerg Infect Dis, 9:1003–1006.
7. Diaz-Quijano FA, Villar-Centeno LA, Martinez-Vega RA. (2005) Effectiveness of early dipyrone administration on severity of dengue virus infection in a prospective cohort. Enferm Infecc Microbiol Clin, 23:593–597.
8. Diaz-Quijano FA, Villar-Centeno LA, Martinez-Vega RA. (2010) Predictors of spontaneous bleeding in patients with acute febrile syndrome from a dengue endemic area. J Clin Virol, 49:11–15
9. Chaterji S, Allen Jr JC, Chow A, Leo YS, Ooi EE. (2011) Evaluation of the NS1 rapid test and the WHO dengue classification schemes for use as bedside diagnosis of acute dengue fever in adults. Am J Trop Med Hyg.;84(2):224–8.
10. Buonora SN, Passos SRL, do Carmo CN, Quintela FM, de Oliveira DNR, dos Santos FB, Hökerberg YHM, Nogueira RMR and Daumas RP. (2016).Accuracy of clinical criteria and an immunochromatographic strip test for dengue diagnosis in a DENV-4 epidemic. BMC Infectious Diseases 16:37
11. Tin Myo Han; Tin Tin Hla; Than Soe; Win Zaw; Tin Aye; Khine Soe Win; Swe Swe Latt; (2016) D.M Thuraiappah Dengue diagnostic tests used by Myanmar General Practitioners as point of care testing 62nd Myanmar Medical Conference. p107.
12. Phuong H L, de Vries PJ, Nga1 TTT, Giao PT, Hung LQ, Binh TQ, Nam NV, Nagelkerke N and Kager PA. (2006) Dengue as a cause of acute undifferentiated fever in Vietnam. BMC Infectious Diseases, 6:123.
13. Oo PM, Wai KT, Harries AD, Shewade HD, Oo T, Thi A, et al. The burden of dengue, source reduction measures, and serotype patterns in Myanmar, 2011 to 2015-R2. Trop Med Health. 2017;45:35.
14. Thu HM, Aye KM & Thein S. (1998) The effect of temperature and humidity on dengue virus propagation in Aedes aegypti mosquitos. Southeast Asian J Trop Med Public Health. Jun: 29(2): 280-4.
15. KT Wai, PT Htun, T Oo, H Myint, Z Lin. Kroeger A, Sommerfeld J, Petzold M. (2012) Community-centred eco-bio-social approach to control dengue vectors: an intervention study form Myanmar. Pathogens and global helath 106(8), 461-468.
16. Sharp TM, Hunsperger E, Santiago GA, Munoz-Jordan JL, Santiago LM, Rivera A, et al. (2013) Virus-specific differences in rates of disease during the 2010 Dengue epidemic in Puerto Rico. PLoS neglected tropical diseases.7(4):e2159.
17. Bodinayake CK, Tillekeratne LG, Nagahawatte A, Devasiri V, Kodikara Arachchi W, Strouse JJ, et al. (2018) Evaluation of the WHO 2009 classification for diagnosis of acute dengue in a large cohort of adults and children in Sri Lanka during a dengue-1 epidemic. PLoS Negl Trop Dis 12(2): e0006258.
18. Daumas RP, Passos SR, Oliveira RV, Nogueira RM, Georg I, Marzochi KB, et al. (2013) Clinical and laboratory features that discriminate dengue from other febrile illnesses: a diagnostic accuracy study in Rio de Janeiro, Brazil. BMC Infect Dis.13:77.
19. Gregory CJ, Lorenzi OD, Colon L, Garcia AS, Santiago LM, Rivera RC, et al. (2011). Utility of the tourniquet test and the white blood cell count to differentiate dengue among acute febrile illnesses in the emergency room. PLoS Negl Trop Dis;5(12), e1400.
20. Huang SY, Lee IK, Wang L, Liu JW, Hung SC, Chen CC, et al. (2014) Use of simple clinical and laboratory predictors to differentiate influenza from dengue and other febrile illnesses in the emergency room. BMC Infect Dis;14:623.
21. Daumas RP, Passos SRL, Oliveira RVC, Nogueira RMR, Georg I, Marzochi KBFand Brasil P. (2013) Clinical and laboratory features that discriminate dengue from other febrile illnesses: a diagnostic accuracy study in Rio de Janeiro, Brazil. BMC Infectious Diseases, 13:77.
22. Potts JA and Rothman AL. (2008) Clinical and laboratory features that distinguish dengue from other febrile illnesses in endemic populations. Trop Med Int Health. November ; 13(11): 1328–1340. doi:10.1111/j.1365-3156.2008.02151.
23. National Guideline for Clinical Management of Dengue.(2018) Vector Borne Disease Conrol Programme, Department of Public Health and Sports, The Republic of the Union of Myanmar. June.
24. Chaterji S , Allen Jr. JC , Chow A , Leo YS , and Ooi EE. (2011) Evaluation of the NS1 Rapid Test and the WHO Dengue Classification Schemes for Use as Bedside Diagnosis of Acute Dengue Fever in Adults. Am. J. Trop. Med. Hyg., 84(2), pp. 224 -228.
25. Gan VC, Lye DC, Thein TL, Dimatatac F, Tan AS, Leo YS. (2013) Implications of discordance in world health organization 1997 and 2009 dengue classifications in adult dengue. PLoS One.8(4), e60946.
25. Buonora SN, Passos SRL, do Carmo CN, Quintela FM, de Oliveira DNR, dos Santos FB, Hökerberg YHM, Nogueira R MR and Daumas RP. (2016) Accuracy of clinical criteria and an immunochromatographic strip test for dengue diagnosis in a DENV-4 epidemic. BMC Infectious Diseases 16:37
Acknowledgement
We would like to acknowledge the General Practitioners who gave permission to conduct this study in their clinics.
Win Lai May1, Aye Aye Win2, Myo Myo Mon3, Han Win4, Kyaw Zin Wai5
1. MBBS, MMedSc (Paediatrics), PhD(Clinical Tropical Medicine), DipRMRE, Department of Medical Research
2. MBBS, MMedSc (Pathology), PhD(Pathology), Department of Medical Research
3. MBBS, MPH, PhD (Epidemiology), Department of Medical Research
4. MBBS, MMedSc(Internal Medicine), FRCP, Department of Medical Research
5. MBBS, MMedSc(Paediatrics), MRCPCH, Department of Paediatrics, University of Medicine 2





