Rabies in Myanmar

Introduction
On the cover of the publication of papers presented at the Third International Symposium on Rabies in Asia (1 Dodet and Meslin, 1997) are pictured beautiful flowers of the Malabar silk-cotton tree in Thailand, known in Myanmar as the “Letpan” tree (Fig. 1A).

Fig. 1A. Cover of Dodet, B, Meslin, F-X. Rabies Control in Asia. Third International Symposium on Rabies Control in Asia. 11-15 September 1996 Wuhan, China. Paris, Elsevier, 1997.
The caption reads: “The time for a dog to become mad is when the Letpan tree (Salmaliamalabarica) (now Bombax ceiba) blossoms. Burmese proverb”. An explanation was offered by Dr Chantapong Wasi (Mahidol University, Bangkok, Thailand). “The mating season for dogs in Asian countries is November-December. As dogs are frequently fighting during this period and as the incubation period for rabies is 1-3 months, they would transmit the virus to each other and develop the disease during summertime, ie, during the months of March and April, which also correspond to nursing of the puppies. This would explain why the number of persons bitten by rabid dogs during summertime increases.” However, in Myanmar, the Letpan tree flowers from mid-January to the end of February (Tabodwe in the Burmese calendar). The flowers attract insects and hence birds, especially flocks of loudly-twittering Burmese mynahs (Acridotheres burmannicus), as I saw myself when travelling with Professor Nyunt Thein near Taunggyi on the 20th January 2015 (Fig. 1B).

Fig. 1B.Burmese mynah (Acridotheres burmannicus) feeding on Malabar silk-cotton (“Letpan”) tree (Bombax ceiba formerly Salmaliamalabarica) near Taunggyi, 20th January, 2015
This behaviour is the origin of the Burmese saying that people talking noisily are “like mynah birds on a Letpan tree”. The Burmese proverb linking the season of canine madness to Letpan flowering was quoted as evidence that rabies had been present in Myanmar since ancient times (Daw Hpay, M., Moneim El-Zawahry, U Saw Ba Heng, Daw Ohn Kyi, Ronald Sein Dwe, U Than Sein and Daw Lily Tong. Some epidemiological features of human rabies and animal bites in Rangoon. Paper presented at the Fifth Burma Research Congress, 7th -11th April, 1970. Rangoon [unpublished], quoted by 2 Bisseru, 1972).
Similarly, in England, there was a popular belief from the Middle Ages, that the hottest part of summer (“dog days” or “canicular days”) associated in ancient times with the heliacal rising of the Dog Star in the Mediterranean area) was the season when dogs were most liable to go mad and inflictrabid bites. One high-profile person is known to have died of rabies in Myanmar in 1910, and in Thailand in 1911(Fig. 2), when Pasteur’s vaccine was available only in Saigon. The Pasteur Institute Burma was established in Rangoon in 1915, allowing confirmation of rabies diagnosis in humans and animals, and production of human and animal vaccines and serum. It became the National Health Laboratory in 1963 (2 Bisseru, 1972).

Admiral Shwe-Maung died of rabiesPrincess Banlusirisarn died of rabies in Bangkok in 1911
in Rangoon in 1910
Fig. 2 Deaths of two high-profile people from rabies in Myanmar in 1910, and in Thailand in 1911, 25 years after Pasteur’s discovery of rabies post-exposure vaccination, yet neither could receive it.
Lyssaviruses
Lyssa viruses are the genus of rabies-related viruses within the family Rhabdoviridae (rod-shaped viruses). Six of the 17 known Lyssaviruses can cause clinical rabies in human beings. These include classic rabies virus (species or genotype 1) which occurs widely in terrestrial mammals and bats in Europe, Africa, and Asia; and in the Americas where it is the only species. Europe has two bat Lyssaviruses (species 5 and 6); in Africa, Duvenhage virus (species 4), named after its first human victim in South Africa, has also been found in insectivorous bats; and Australia has its own bat Lyssavirus (species 7). The recently discovered IrkutLyssavirus (species 10) was found in bats and dogs in China and the Russian Far East and has been responsible for a few human fatalities.
Epidemiology
The epidemiology of rabies, the classic zoonosis, is summarised in Fig 3. In the sylvatic (wildlife) cycle, strains of virus tend to circulate mainly within a particular wild mammalian species, such as foxes in Europe, but infection across species may occur, for example if a wild or domestic carnivore attacks a sick bat. The urban cycle involves domestic dogs and cats (increasingly feral cats in North America), whose vast numbers (dog: human population ratio 1:8 in Sri Lanka, 1:6 in Myanmar, 1:3.6 in USA), and intimate relationship to humans, poses the highest risk of rabies in most countries.
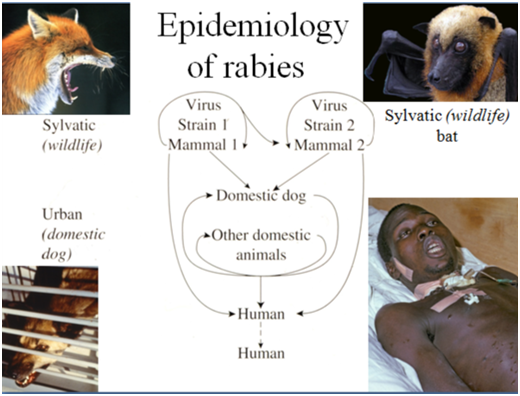
Fig.3 Epidemiology of rabies in wildlife and domestic dogs
WHO estimated that, out of a global total of 59,000 human deaths from rabies, there were 35,172 deaths per year in Asia, of which India accounted for 60% in Asia and 35% globally (3 WHO, 2018) (Fig 4.).
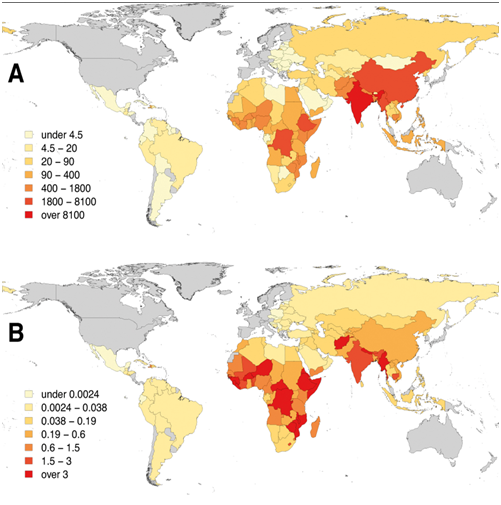
Fig. 4A: Human deaths from rabies; B: Death rates per capita (per 100,000 population); countries shaded in grey are free from canine rabies (reference 3 WHO Expert Consultation on Rabies: WHO TRS N°1012 2018).
At a meeting of the Myanmar Medical Association in January 2018, I learnt from Dr Tin Tin Kyi that, according to the Ministry of Public health, there had been more deaths from rabies than malaria in Myanmar since 2014. This resulted from the steep decline in malaria deaths since 2009, so that, in 2015, they were 0.2 rabies deaths/100,000 population/year, compared to 0.1 malaria deaths/100,000 population (Fig. 5).
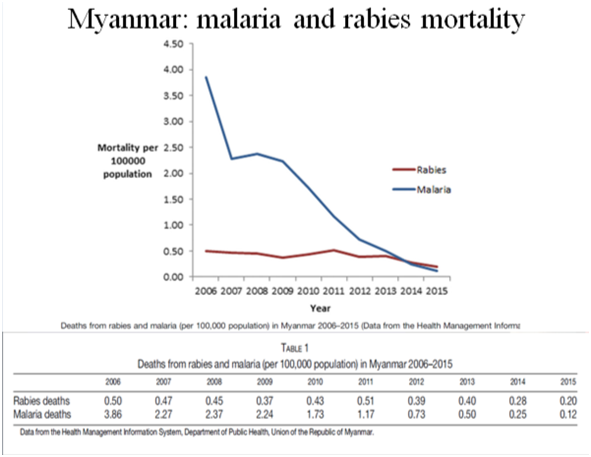
Fig. 5 Malaria and rabies mortalities in Myanmar, 2008-2015
In Myanmar, there are about 600,000 dog bites a year and 1,000 human rabies deaths per year, whereas in India, there are 17½ million dog bites a year resulting in about 20,000 human deaths (1.8/100,000) despite 8 million post-exposure courses, and in Bangladesh 300,000 dog bites a year resulting in 2,500 human deaths (1.3/100,000). In India, a direct estimate of human deaths with symptoms of classic furious rabies provided by the million-death study organised by the Registrar General of India was 12,700 deaths in a 2005 (99%, confidence intervals 10,000 – 15,500) (4 Suraweera et al. 2012).
In Myanmar, domestic dogs including strays are the most important reservoir and vectors for human rabies. Domestic cats are responsible in a few cases. A study of the molecular epidemiology of dog rabies identified a lineage SEA3 confined to dogs from Myanmar, distinct from lineages SEA1 and SEA2 that were prevalent in Cambodia and other SE Asian countries (5 Mey et al., 2016). There is a possibility of spill-over of rabies virus to domestic dogs and cats from wildlife and vice-versa. Monkeys, horses, cattle, goats, rabbits, rats have been mentioned as being occasionally infected with rabies in Myanmar (6 Ye-Aye, 1997; 7Than-Hla, 2001). However, other wild mammal species that have been implicated as rabies reservoirs in Bangladesh, India and Nepal (8 Gongal, 2006) such as mongoose (Herpestes spp.), jackal (Canis aureus), fox (Vulpes bengalensis), wolf (Canis lupus), and bats, have not been investigated in Myanmar (9 Myanmar Ministry of Agriculture, Livestock and Irrigation, 2018). Some new mammalian viruses have been discovered in bats from Myanmar by Chinese investigators in bordering Yunnan Province, but, so far, no Lyssavirus has been identified. Whether bats in Myanmar carry rabies viruses, as they do in Thailand, China, India, and the Philippines, remains an important but still unanswered question. Bats are increasingly implicated as vectors and reservoirs of human diseases, including COVID-19 (10 Tian et al., 2022). Immense clouds of bats are a familiar sight in some parts of Myanmar, for example at dusk around Than Lwin Bridge, Hpa An, in Kayin State (Fig. 6).

Fig. 6 Cloud of bats at Than Lwin Bridge, Hpa An, Kayin State, Myanmar. Might they be carrying rabies virus?
Clinical disease in dogs
Furious rabies in dogs, with classic features of canine madness, is easily recognised. The animal becomes aggressive, attacking people, other animals, and even inanimate objects, and wandering from home. But only a minority of rabid dogs (40%) show this clinical pattern. The majority (60%) develop “dumb” or paralytic symptoms, drooling saliva, losing their bark, showing paralysis of the neck muscles, and hind legs, and cowering under the furniture at home. Rabid domestic cats nearly always have furious symptoms.
Human rabies: human-to-human transmission after tissue/organ transplantation
Except through acquisition via tissue/organ transplants, human-to-human transmission of rabies virus has never been confirmed since virological confirmation became possible, even though virus is present in the saliva and other secretions of human victims who may be nursed by family members at home. Recipients of corneal and solid organ (liver, kidney, iliac artery) grafts have died of rabies. All the donors had died of undiagnosed neurological illnesses, but it was later confirmed that they had, in fact, been suffering from rabies encephalomyelitis (11 Lu et al., 2018).
Pathogenesis of rabies encephalomyelitis
Rabies virus, inoculated by bites or scratches in the saliva of the rabid mammal, may multiply locally in muscle cells for short time before gaining access to the nervous system mainly by binding to acetylcholine receptors at the neuro-muscular junctions of intrafusal fibres of muscle spindles. This invasion can be blocked experimentally by competitive binding of curariform post-synaptic snake venom toxins such as α-bungarotoxin. Once in the axoplasm, the virus is transported retrograde by the physiological dyne in motor, to the spinal cord where it multiplies in neurones and moves inter-neuronally and via the cerebrospinal fluid (CSF) to the brain where further replication occurs. There is then centrifugal spread of virus via peripheral nerves to salivary and lacrimal glands, skin, heart, lungs and other organs and tissues. In nature, this allows infection of other hosts by virus excreted in saliva, facilitated by the mood change to aggression in the infected mammal.
Human rabies encephalomyelitis
The incubation period is usually a few months, but in exceptional cases may be up to a documented 19 years, suggesting latency as with Herpes viruses. The earliest prodromal symptom is usually itching at the site of the by-now-healed bite wound. This may be intense enough to provoke scratching with excoriation. A variety of diagnostically-misleading non-specific physical or psychological prodromal symptoms may suggest respiratory, gastro-intestinal or emotional aetiologies. After a few days, overt disease develops, either furious rabies, with pathognomonic hydrophobic spasms, autonomic over-activity, and rapid deterioration into coma and death over a few days; or more insidious paralytic (“dumb”) rabies with ascending flaccid paralysis without hydrophobia, and survival for up to 30 days even without supportive care (12 Warrell, 1976). Hydrophobic spasms are jerky inspiratory muscle spasms, provoked by attempts to swallow or, through conditioning, by the mere sight of water or sound of a running tap (13 Warrell et al., 1976). The same reflex is stimulated by a current of air against the skin, “aerophobia”. In both cases, there is associated, indescribable terror. Patients may fluctuate between periods of extreme arousal and even aggression, unconsciousness, and lucid intervals during which they may be aware of their awful predicament.
Confirmation of diagnosis
Demonstration of intra-cytoplasmic inclusions Negri bodies, composed of masses of rabies virions, by Seller’s stain, the classic method, has been replaced by more sensitive and specific methods, such as RT-PCR using saliva, CSF, tears, or skin biopsy; virus isolation from saliva, brain, CSF, or urine, during the first week of illness; immune fluorescent detection of rabies antigen in skin punch biopsies (14 Bryceson et al., 1975; 15Warrell et al., 1988); neutralising antibody response in serum and CSF; and autopsy or post-mortem needle necropsy of brain.
Prognosis of human rabies encephalomyelitis
This is the most highly fatal of all infectious diseases. There is no specific treatment. However, two children bitten by insectivorous bats in the USA in 1970 and 2005, one of whom had received no vaccine, made virtually complete recoveries, while a number of incompletely vaccinated or unvaccinated patients in India, South Africa, Mexico, Argentina, Brazil (bat), and USA have survived, albeit with severe neurological sequelae. Dog-transmitted rabies has proved 100% fatal in unvaccinated patients.
Treatment of rabies encephalomyelitis
Because of this bleak prognosis, only in exceptional circumstances should intensive care be considered. Unfortunately, the “Milwaukee Protocol” (tetrahydrobiopterin, ketamine, amantadine, favipiravir, sapropterin and L-arginine or nimodipine, and vitamin C), improvised by Willoughby to treat one of the only two patients to recover from rabies encephalomyelitis (16 Willoughby et al., 2005), has proved ineffective in all subsequent cases (17 Zeiler and Jackson, 2016). In many clinics and hospitals in rabies-endemic areas of the world, patients suspected of having rabies are considered untreatable and are either sent home to die with relatives without advice, or are isolated and abandoned in a remote part of the hospital and denied medical attention. These practices ignore the fundamental precept that a doctor’s responsibility is to relieve suffering even if there is no possibility of cure. In every case of rabies encephalomyelitis, palliation of pain, terror and distress is imperative. Ideally, patients should be accommodated in calm, quiet conditions, allowing their relatives to communicate with them in safety and privacy (18 Warrell et al., 2017).
Prevention
Survival from rabies encephalomyelitis is extremely rare, but, fortunately, this terrible disease is eminently preventable through personal protection (pre- and post- exposure prophylaxis), and, at a public health level, by control of canine and wild-life rabies.
Personal protection
There are two strategies – primary post-exposure prophylaxis (PEP) and Pre-exposure prophylaxis (PrEP) followed by post-exposure boosting if and when required (Fig. 7).
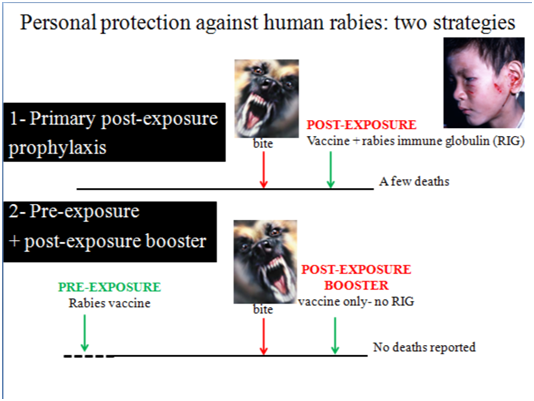
Fig. 7 The two strategies for personal protection against rabies: Primary post-exposure prophylaxis (PEP) and Pre-exposure prophylaxis (PrEP) followed by Post-exposure boosting after subsequent exposure
Primary post-exposure prophylaxis
This is a medical emergency demanding: 1-thorough wound cleaning preferably with soap under a running tap and then application of iodine or alcohol; 2-passive immunisation with rabies immune globulin (human or equine RIG) infiltrated round the bite or scratch wounds; and 3-a course of active immunisation, using a modern tissue/cell culture vaccine.
Passive immunisation with RIG provides ready-made neutralising antibody in the area where virus has been inoculated, to kill the virus during the first 7 days after starting vaccination, before the response to active immunisation has reached protective levels. It is recommended for HIGH-RISK EXPOSURES, defined as: “bites or scratches that break the skin causing bleeding; contact of animals’ saliva with broken skin or intact mucous membrane; or any direct contact with a bat”. In the case of immune-compromised patients, RIG is also given for LOW-RISK EXPOSURES, defined as: “bites or nibbles without breaking skin; minor scratches or abrasions – without bleeding”. The dose of RIG is strictly according to body weight and should not be exceeded. It is infiltrated around all the bite wounds as soon as possible and may have to be diluted if wounds are numerous. Adequate anaesthesia, even general anaesthesia, may be necessary in children. Inject vaccine as far away from the wound as possible to minimise the risk of cross-neutralisation.
The standard (“Essen”) vaccination regimen, recommended in North America and Europe, involves doses of 1 vial (0.5 or 1.0 ml depending on the type of vaccine) intramuscularly, given on days 0, 3, 7, and on day 14-21, with RIG on Day 0 for high-risk exposures. Immuno-suppressed patients receive a 5th dose on day 28-30 (see below). This regimen is far too expensive for most of the low-middle income countries where rabies is most prevalent. This inequity prompted Dr Mary Warrell and her team at Queen Saovabha Memorial Institute (Thai Red Cross), in Bangkok to investigate alternative, economical, post-exposure regimens. In human volunteers, various routes and adjuvants were tested, but multi-site, intradermal vaccination proved the most rapidly immunogenic, producing levels of rabies neutralising antibody at least as high as those in people receiving the standard intramuscular regimen (19 Warrell et al.,1983). Intra-dermal immunisation is probably more immunogenic because dermal dendritic cells convey vaccine antigens to the reticulo-endothelial system, while multi-site immunisation recruits more groups of regional lymph nodes to neutralising antibody production. The multi-site intradermal regimen proved effective in patients bitten by confirmed-rabid dogs in Bangkok (20 Warrell et al., 1985), and was approved by WHO (Fig. 8). An alternative regimen [“Thai Red Cross (TRC)”] has also been promoted. However, the most economical regimen, especially for smaller clinics, is a 2 visit, 4-site intradermal, one-week, post-exposure regimen (21 Warrell, 2019A; 22Warrell 2019B).
Fig 8. First recommendation by WHO of economical intradermal use of rabies vaccines in 1997.
WHO recommendations on rabies post-exposure treatment and the correct technique of intradermal immunization against rabies World Health Organization. (1997) Document numberWHO/EMC/ZOO/96.6https://apps.who.int/iris/handle/10665/63396
Failures of rabies post-exposure prophylaxis
Rabies post-exposure prophylaxis has saved countless lives since it was introduced by Louis Pasteur in 1885 (23 Pearce, 2002). However, a number of failures of PEP have been reported, attributable to delay in treatment, exceptionally severe multiple bites, suboptimum methods of vaccination, or omission of passive immunisation, and sometimes, impairment of the host’s immune response 24 (Lancet, 1988). These failures usually occurred in low-resource settings. However, a recent case was reported in USA (25 Holtzbauer et al., 2023). An 84-year-old man was bitten on his hand by a bat. Full PEP was started 3 days later when rabies was confirmed in the bat. Five months later, he developed symptoms of paralytic rabies from which he died after 15 days’ illness. Absence of detectable rabies neutralising antibody in his serum and CSF indicated that there had been no response to the PEP given 5 months previously. This was later explained by an unsuspected IgM monoclonal gammopathy, secondary to prostatic adenocarcinoma. The authors considered that host-mediated primary vaccine failure due to immune dysfunction was the most likely explanation for PEP failure. They suggest that clinicians should consider measuring rabies neutralizing antibody titres after completion of PEP if there is any suspicion of immune-compromise. They admitted that the 3-day delay in giving RIG might have contributed to failure of PEP. For bat bites, WHO recommends that rabies vaccine and RIG should be administered immediately (WHO, 2018).
Pre-exposure prophylaxis followed by post-exposure boosting when required
This is recommended for residents of, and travellers to dog rabies enzootic areas, laboratory workers dealing with rabies virus, animal handlers, and staff of hospitals where patients with rabies are admitted frequently. WHO recommends a course of two injections (one site 1.0/0.5 ml intramuscular; or two site 0.1 ml intradermal) on days 0 and 7. CDC (USA) and HSA (UK) recommend a third dose on day 21-28. Booster doses are not necessary for those living in, or travelling to, high-risk areas who have already received a primary series of pre- or post- exposure rabies prophylaxis. However, for those with persistent and frequent high risk, a routine booster dose after 1-2 years is recommended with or without checking the serum neutralising antibody titre. WHO considers that mass pre-exposure vaccination for entire populations is not cost–effective and therefore not recommended, except in remote areas with limited access to post-exposure vaccination, if the annual dog bite incidence is more than 5%, or for indigenous communities in South America exposed to vampire bats (WHO, 2018)
Post-exposure boosting of those, previously-vaccinated, who are subsequently exposed
A single visit, four-site intradermal injection of 0.1 ml at each site is recommended. This regimen has proved more immunogenic than two visits on days 0 and 3, to receive 1.0/0.5 ml at one site intramuscularly or 0.1 ml intradermally at each of two sites. No failures have been reported of this strategy of pre-exposure vaccination followed by a post-exposure booster, a remarkable record for such a dangerous infection.
Rabies post-exposure prophylaxis: common queries
1-When is it too late to give post-exposure prophylaxis? – Due to the potentially long and unpredictable incubation period of rabies there is no time limit for giving post-exposure prophylaxis – risk in every case of potential exposure should be assessed individually.
2-When is it too late to give rabies immune globulin (RIG)? – If the exposure was more than one year previously, RIG is no longer indicated
3-When is it too late to give RIG after starting vaccine? – RIG is not given more than 7 days after starting rabies post-exposure vaccination, or to immune competent people who have already been partially or previously immunised
4-What about the health of the dog responsible for the bite? – If the biting dog remains well and behaves normally 15 days after the attack it is not rabid (but never delay starting post-exposure prophylaxis until then!)
Controlling rabies in domestic and wild mammals
Urban rabies: owned dogs should be muzzle, or restricted to the owner’s compound. Strays should be caught, vaccinated, sterilised and then released. Domestic dog vaccination campaigns have successfully eliminated human rabies in several large cities in South America, and reduced the incidence of human rabies (e.g. in Sri Lanka Fig. 9)
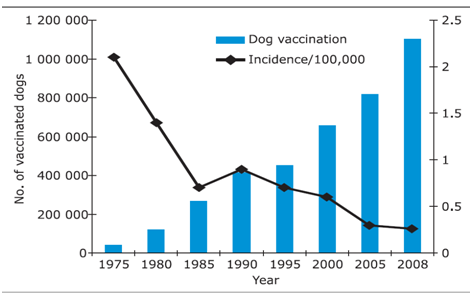
Fig. 9 Impact of the mass dog vaccination campaign on human rabies incidence in Sri Lanka, 1975-2008
In November 2018, the Myanmar Ministry of Agriculture, Livestock and Irrigation published its NATIONAL ACTION PLAN FOR RABIES ELIMINATION IN DOG 2018-2030, which, in parallel with Global and ASEAN strategies, aims to eliminate canine rabies in Myanmar by 2030. Myanmar’s Veterinary Services will be strengthened to carry out mass dog vaccination, and improve rabies surveillance, diagnosis, research, and dog population control. The plan consists of socio-cultural, technical, organizational, political and resources components (9 Myanmar Ministry of Agriculture, Livestock and Irrigation, 2018).
Wildlife rabies: distribution of oral live-attenuated vaccine in baits has achieve 50-75% herd immunity and eliminated wildlife rabies in several countries (e.g. in red foxes in Western Europe).
Rabies-free countries (islands) should consider quarantine and vaccination of imported domestic dogs
Conclusions
Rabies encephalomyelitis is incurable (although patients deserve full palliative treatment), but it is readily preventable and controllable.
The modern tissue/cell culture vaccines used for pre- and post-exposure prophylaxis are potent, safe, very effective but expensive.
Multi-site intradermal administration of rabies vaccine is safe, effective and economical.
Rabies immune globulin (RIG) is recommended for immediate passive immunisation after severe/high risk exposures, but is unavailable or unaffordable in many countries.
Immuno-suppressed patients require an additional booster dose of vaccine, and RIG even for low-risk exposure.
Pre-Exposure Prophylaxis should be considered by residents of, or travellers to dog rabies enzootic areas.
Post-Exposure boosters are essential for those who have had pre-exposure vaccination and are subsequently exposed, but RIG is unnecessary
Control of canine rabies is the most economical method of preventing human rabies.
References
- Dodet, B, Meslin, F-X. Rabies Control in Asia. Third International Symposium on Rabies Control in Asia. 11-15 September 1996 Wuhan, China. Paris, Elsevier, 1997.
- Bisseru B. Rabies. London, William Heinemann Medical Books,1972.
- WHO Expert Consultation on Rabies, third report. Geneva: World Health Organization; 2018 (WHO Technical Report Series, No. 1012).
- Suraweera W, Morris SK, Kumar R, Warrell DA, Warrell MJ, Jha P; Million Death Study Collaborators. Deaths from symptomatically identifiable furious rabies in India: a nationally representative mortality survey. PLoS Negl Trop Dis. 2012;6(10):e1847.
- Mey C, Metlin A, Duong V, Ong S, In S, Horwood PF, Reynes JM, Bourhy H, Tarantola A, Buchy P. Evidence of two distinct phylogenetic lineages of dog rabies virus circulating in Cambodia. Infect Genet Evol. 2016 Mar;38:55-61.
- Ye-Aye. Myanmar Human aspects of rabies prevention and control. In Dodet, B, Meslin, F-X. Rabies Control in Asia. Third International Symposium on Rabies Control in Asia. 11-15 September 1996 Wuhan, China. Paris, Elsevier, 1997: 171-4. Thanh-Binh-Swe, 2001
- Than-Hla. Rabies control in Myanmar. Fourth International Symposium on Rabies Control in Asia. Organized by the Merieux Foundation with co-sponsorship of the World Health Organization. 5-9 March 2001, Hanoi, Vietnam.pp 55-7.
- Gongal GN. The epidemiological trend of animal rabies in Nepal and future control strategy. Journal of the Association for Prevention and Control of. Rabies in India. 2006;8(1).
- Myanmar Ministry of Agriculture, Livestock and Irrigation, 2018. THE REPUBLIC OF THE UNION OF MYANMAR Ministry of Agriculture, Livestock and Irrigation Livestock Breeding and Veterinary Department. NATIONAL ACTION PLAN FOR RABIES ELIMINATION IN DOG 2018-2030.
- Tian J, Sun J, Li D, Wang N, Wang L, Zhang C, Meng X, Ji X, Suchard MA, Zhang X, Lai A, Su S, Veit M. Emerging viruses: Cross-species transmission of coronaviruses, filoviruses, henipaviruses, and rotaviruses from bats. Cell Rep. 2022 Jun 14;39(11):110969.
- Lu XX, Zhu WY, Wu GZ. Rabies virus transmission via solid organs or tissue allotransplantation. Infect Dis Poverty. 2018 Aug 15;7(1):82.
- Warrell DA. The clinical picture of rabies in man. Trans R Soc Trop Med Hyg. 1976;70(3):188-95.
- Warrell DA, Davidson NM, Pope HM, Bailie WE, Lawrie JH, Ormerod LD, Kertesz A, Lewis P. Pathophysiologic studies in human rabies. Am J Med. 1976 Feb;60(2):180-90.
- Bryceson AD, Greenwood BM, Warrell DA, Davidson NM, Pope HM, Lawrie JH, Barnes HJ, Bailie WE, Wilcox GE. Demonstration during life of rabies antigen in humans. J Infect Dis. 1975 Jan;131(1):71-4.
- Warrell MJ, Looareesuwan S, Manatsathit S, White NJ, Phuapradit P, Vejjajiva A, Hoke CH, Burke DS, Warrell DA. Rapid diagnosis of rabies and post-vaccinal encephalitides. Clin Exp Immunol. 1988 Feb;71(2):229-34.
- Willoughby RE Jr, Tieves KS, Hoffman GM, Ghanayem NS, Amlie-Lefond CM, Schwabe MJ, Chusid MJ, Rupprecht CE. Survival after treatment of rabies with induction of coma. N Engl J Med. 2005 Jun 16;352(24):2508-14.
- Zeiler FA, Jackson AC. Critical Appraisal of the Milwaukee Protocol for Rabies: This Failed Approach Should Be Abandoned. Can J Neurol Sci. 2016 Jan;43(1):44-51.
- Warrell M, Warrell DA, Tarantola A. The Imperative of Palliation in the Management of Rabies Encephalomyelitis. Trop Med Infect Dis. 2017 Oct 4;2(4):52.
- Warrell MJ, Warrell DA, Suntharasamai P, Viravan C, Sinhaseni A, Udomsakdi D, Phanfung R, Xueref C, Vincent-Falquet JC, Nicholson KG, Bunnag D, Harinasuta T. An economical regimen of human diploid cell strain anti-rabies vaccine for post-exposure prophylaxis. Lancet. 1983 Aug 6;2(8345):301-4.
- Warrell MJ, Nicholson KG, Warrell DA, Suntharasamai P, Chanthavanich P, Viravan C, Sinhaseni A, Chiewbambroongkiat MK, Pouradier-Duteil X, Xueref C, et al.Economical multiple-site intradermal immunisation with human diploid-cell-strain vaccine is effective for post-exposure rabies prophylaxis. Lancet. 1985 May 11;1(8437):1059-62.
- Warrell MJ. Rabies post-exposure vaccination in 2 visits within a week: A 4-site intradermal Vaccine. 2019A Feb 21;37(9):1131-1136.
- Warrell MJ. Simplification of Rabies Postexposure Prophylaxis: A New 2-Visit Intradermal Vaccine Regimen. Am J Trop Med Hyg. 2019B Dec;101(6):1199-1201.
- Pearce JMS. Louis Pasteur and rabies: a brief note. J NeurolNeurosurg Psychiatry 2002 Jul;73(1):82.
- Rabies vaccine failures. Lancet 1988 Apr 23;1(8591):917-8.
- Holzbauer SM, Schrodt CA, Prabhu RM, et al., Fatal Human Rabies Infection with Suspected Host-mediated Failure of Post-Exposure Prophylaxis Following a Recognized Zoonotic Exposure-Minnesota, 2021. Clin Infect Dis. 2023 Mar 29:ciad098. doi: 10.1093/cid/ciad098. Online ahead of print.
Author Information
David A. Warrell,
Nuffield Department of Clinical Medicine,
University of Oxford, UK



