Indetermined biliary obstruction and filling defects have been a clinical dilemma with broad differential diagnoses including malignant lesions. Owing to direct visualization of biliary tree, cholangioscopy becomes a useful tool in this situation. A middle age woman was referred from a remote area of Ayarwaddy division with obstructive jaundice and aching pain in the right hypochondrium. Her abdominal ultrasonography showed biliary tree dilatation due to stone at the proximal end of common bile duct. But there were two different opinions on CT scan: the first opinion was dilated common bile duct with suspicion of periductal infiltrative type of cholangiocarcinoma and second opinion suggested the possibility of hepatolithiasis. Therefore, endoscopic ultrasound was done and it revealed a hypoechoic mass at the common hepatic and proximal left intrahepatic duct. Endoscopic retrograde cholangiopancreatography with subsequent intraductal biopsy reported neoplastic cells which on immunohistochemistry staining favoured leiomyoma. Single operator cholangioscopy showed protruding polypoid lesion with papillary projection at common hepatic duct extending to proximal left intrahepatic duct suggestive of malignancy. Targeted biopsy revealed poorly differentiated cholangiocarcinoma.
Introduction
Regardless of significant advances in pancreaticobiliary imaging and endoscopic ultrasound (EUS) guided assessment of tumors, the characterization of intraductal biliary lesions remains a challenge. Biliary strictures are considered indeterminate if the aetiology is uncertain with inconclusive/negative imaging or tissue diagnosis1.
Accurate and timely diagnosis is important to achieve curative surgical resection or palliative biliary drainage of malignant biliary stricture whereas the treatment of benign biliary strictures relies on medical and endoscopic management with only small proportions requiring surgical intervention. Evaluation of these strictures is challenging and often requires a multidisciplinary approach and multiple procedures. Transabdominal ultrasound, abdominal multi-detector computed tomography, magnetic resonance imaging and magnetic resonance cholangiopancreatography are commonly used as the initial diagnostic workups to demonstrate the existence, location and extent of biliary strictures, to aid in tumor detection and determine its resectability.
Cholangioscopy, which facilitates direct visual assessment and visually guided tissue sampling, holds promise as an advanced technique in cases that elude successful diagnosis by conventional endoscopic retrograde cholangiopancreatography (ERCP) or other imaging modalities2. Recently, single operator cholangioscope with direct visualization system has become available to overcome the limitations of previous methods. The system directly visualizes the biliary tree for diagnostic and therapeutic purposes by using a disposable digital scope with 120 degrees field of view. A tapered tip, a four-way tip deflection system, and 2 dedicated channels for irrigation and an aspiration port, which enabled unrestrained observation and biopsy procedures3.
Case presentation
A 49-year-old woman from Ayarwaddy division was referred to our outpatient department for hepatobiliary surgical department. She had problems of obstructive jaundice and aching pain in the right hypochondrium. She initially consulted a surgeon and investigations were done, including ultrasound abdomen, CT (abdomen) and liver function test (LFT). She was then referred to a tertiary center for further management. On reviewing her investigations ultrasound abdomen showed biliary tree dilatation due to stone in proximal end of common bile duct (CBD). CT scan report by first radiologist identified a segmental irregular dilatation in the left intrahepatic duct (IHD) with diffuse periductal thickening and increased enhancement, CBD is also dilated, with mural enhancement and enhancing mass like density in proximal CBD suspicious of periductal infiltrative type of cholangiocarcinoma. Subsequent opinion by a different radiologist suggested the possibility of hepatolithiasis because the left IHD was dilated, with few hyperdense foci in the left biliary system and common hepatic duct (CHD). She was decided to do EUS as first step before proceeding invasive procedure like ERCP. On EUS examination, hypoechoic mass was identified at CHD and proximal left IHD (Fig 1). Then ERCP was done. Similarly, filling defect was noted at CHD and proximal left IHD on cholangiogram (Fig 2). Intraductal biopsy (Fig 3) taken during ERCP revealed neoplastic cells weakly positive with smooth muscle actin which favouring leiomyoma on immunohistochemistry (IHC) staining. Cholangioscopy (SpyGlass DS, Boston Scientific, USA) showed a protruding polypoid lesion with some papillary projection,some ulcers and haemorrhagic areas in biliary confluence extending to proximal left IHD(Fig 4a and 4b). Targeted biopsy with the SpyBite forceps (Boston Scientific, USA) reported poorly differentiated cholangiocarcinoma (Fig 5).
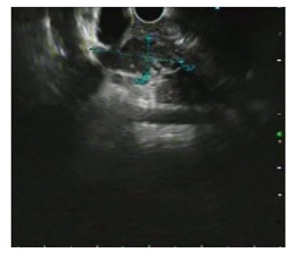
Fig 1– EUS showing hypoehoic mass at the Common Hepatic Duct
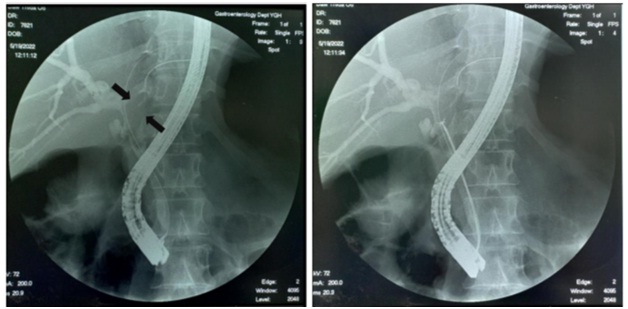
Fig 2- Cholangiography showing filling defect Fig 3- Intraductal biopsy taken from filling defect in the CHD and proximal left IHD (arrow) of CHD and proximal left IHD
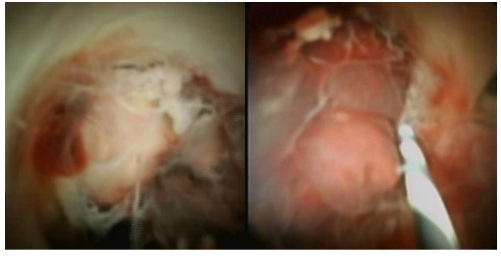
Fig 4a and 4b– Single operator cholangioscopy (Spyglass) showing protruding polypoid lesion with some papillary projection,some ulcers and haemorrhagic areas in biliary confluence extending to proximal left IHD.
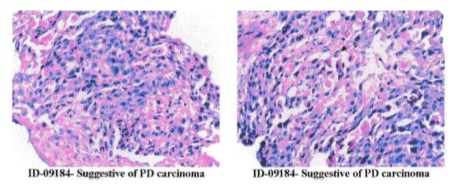
Fig 5- Cholangioscope guided biopsy showing large cluster of atypical polygonal cells which has hyperchromatic nuclei with increase Nuclear-cytoplasmic ratio and high mitotic activity. Attenuated gland formation or vasoformative patterns are noted
Discussion
Biliary lesion has been considered as indeterminate after a definite diagnosis cannot be achieved by standard approaches (i.e., USG/EUS/ ERCP) tools. Up to 20% of biliary strictures remain indeterminate after the standard approach and these patients underwent surgery to achieve a definitive diagnosis in the past. Cholangioscopy allows access and visualization of the biliary tree and procedures can be performed via the percutaneous, transhepatic or per-oral routes. Diagnostic cholangioscopy is mainly required for the evaluation of biliary strictures but can also be useful to detect aetiology of unexplained filling defects at fluoroscopy and abnormalities detected during CT, MRI and EUS. Moreover, its application can help defining longitudinal extension of cholangiocarcinoma (CCA) more accurately than the fluoroscopic measurement during ERCP 4.
Our case was considered to be indeterminate because EUS examination revealed hypoechoic mass at CHD and proximal left IHD and ERCP with intraductal biopsy showed neoplastic cells which were weakly positive with smooth muscle actin on IHC staining favouring leiomyoma. Cholangioscopy was proceeded for more definitive diagnosis as leiomyoma in biliary tree is very rare.
A systematic review reported that four characteristics (tumor vessels, papillary projection, nodular or polypoid mass, and infiltrative lesions) were commonly used for detection of malignant/neoplastic biliary strictures at cholangioscopy 5. An overall sensitivity of 94% was reported for the visual diagnosis of malignant/neoplastic biliary strictures in a systematic review and a meta-analysis5,6. Historically, neovascularization, also termed “tumor vessels,” has had the most consensus regarding its description and malignant implications 7. It has been described as irregularly dilated, tortuous, and abnormally proliferating vessels on the mucosa adjacent to a stricture. Based on their study, in our case, SpyGlass DS demonstrated a protruding polypoid lesion with some papillary projection,some ulcers and haemorrhagic areas which favored malignancy.
In addition to the visual diagnosis of intraductal biliary strictures (IDBSs), peroral-cholangioscopy guided biopsy can provide further histopathologic interpretation of IDBSs. In a systematic review with metaanalysis of 10 studies evaluating the use of single operator cholangioscopy (SOC)- guided biopsy for the diagnosis of malignant biliary strictures, the overall pooled sensitivity and specificity were 60.1% and 98.0% respectively 8. SOC-guided biopsy of our patient reported as poorly differentiated adenocarcinoma which was compactable with cholangioscopic visual impression.
Although there were no obvious adverse events in our case, the overall adverse events (AEs) rate of the single-operator cholangiopancreatoscope (SOCP) for both diagnostic and therapeutic procedures was between 2% and 30%. The AEs included cholangitis, pancreatitis, haemobilia, bile leak. Air embolization and bile duct perforation were rare and serious complications9.Complication rates of pancreatitis and perforation are comparable to ERCP, however, cholangitis rate was significantly higher in cholangioscopy (1% vs 0.2% in ERCP)10.
Conclusion
Cholangioscopy becomes a useful procedure due to its direct visualization of biliary tree and therapeutic intervention in indeterminate biliary lesion while standard approaches tools failed to give definitive diagnosis.
References
- Angsuwatcharakon, P., Kulpatcharapong, S., Moon, J. H., Ramchandani, M., Lau, J., Isayama, H., Seo, D. W., Maydeo, A., Wang, H. P., Nakai, Y., Ratanachu-Ek, T., Bapaye, A., Hu, B., Devereaux, B., Ponnudurai, R., Khor, C., Kongkam, P., Pausawasdi, N., Ridtitid, W., Piyachaturawat, P., … Rerknimitr, R. (2022). “Consensus guidelines on the role of cholangioscopy to diagnose indeterminate biliary stricture. HPB”: the official journal of the International Hepato Pancreato Biliary Association, 24(1), 17–29.
- Ramchandani, M., Reddy, D. N., Gupta, R., Lakhtakia, S., Tandan, M., Darisetty, S., Sekaran, A., & Rao, G. V. (2011). “Role of single-operator peroral cholangioscopy in the diagnosis of indeterminate biliary lesions: a single-center, prospective study.” Gastrointestinal endoscopy, 74(3), 511–519.
- Karagyozov, P., Boeva, I., & Tishkov, I. (2019). “Role of digital single-operator cholangioscopy in the diagnosis and treatment of biliary disorders”. World journal of gastrointestinal endoscopy, 11(1), 31–40.
- Ghisa, M., Bellumat, A., De Bona, M., Valiante, F., Tollardo, M., Riguccio, G., Iacobellis, A., Savarino, E., & Buda, A. (2022). “Biliary Tree Diagnostics: Advances in Endoscopic Imaging and Tissue Sampling”. Medicina (Kaunas, Lithuania), 58(1), 135.
- Kulpatcharapong, S., Pittayanon, R., J Kerr, S., & Rerknimitr, R. (2020). “Diagnostic performance of different cholangioscopes in patients with biliary strictures: a systematic review.” Endoscopy, 52(3), 174–185.
- de Oliveira, P. V. A. G., de Moura, D. T. H., Ribeiro, I. B., Bazarbashi, A. N., Franzini, T. A. P., Dos Santos, M. E. L., Bernardo, W. M., & de Moura, E. G. H. (2020). “Efficacy of digital single-operator cholangioscopy in the visual interpretation of indeterminate biliary strictures: a systematic review and meta-analysis”. Surgical endoscopy, 34(8), 3321–3329.
- Kim, H. J., Kim, M. H., Lee, S. K., Yoo, K. S., Seo, D. W., & Min, Y. I. (2000). “Tumor vessel: a valuable cholangioscopic clue of malignant biliary stricture.” Gastrointestinal endoscopy, 52(5), 635–638.
- Navaneethan, U., Hasan, M. K., Lourdusamy, V., Njei, B., Varadarajulu, S., & Hawes, R. H. (2015). “Single-operator cholangioscopy and targeted biopsies in the diagnosis of indeterminate biliary strictures: a systematic review.” Gastrointestinal endoscopy, 82(4), 608–14.e2.
- Parsa, N., & Khashab, M. A. (2019). “The Role of Peroral Cholangioscopy in Evaluating Indeterminate Biliary Strictures”. Clinical endoscopy, 52(6), 556–564.
- Sethi, A., Chen, Y. K., Austin, G. L., Brown, W. R., Brauer, B. C., Fukami, N. N., Khan, A. H., & Shah, R. J. (2011). “ERCP with cholangiopancreatoscopy may be associated with higher rates of complications than ERCP alone: a single-center experience.” Gastrointestinal endoscopy, 73(2), 251–256.
Author Information
Than Than Aye1 , Ohnmar Nyunt Tin2 , Swe Mon Mya3 ,Sandar Win4 , Tin Moe Wai5 , Mya Thet Nwe6 , Thida Soe7
- Professor & Head, Department of Gastroenterology, University of Medicine 1, Yangon.
- Consultant Gastroenterologist, Department of Gastroenterology, Yangon General Hospital.
- Senior Consultant Gastroenterologist, Department of Gastroenterology, Yangon General Hospital.
- Ass: Professor, Department of Gastroenterology, University of Medicine 1, Yangon.
- Ass: Professor, Department of Gastroenterology, University of Medicine 1, Yangon.
- Senior Consultant Gastroenterologist, Department of Gastroenterology, Yangon General Hospital.
- Consultant Gastroenterologist, Department of Gastroenterology, Yangon General Hospital.






