Abstract
Venous thromboembolism including pulmonary embolism (PE) and deep vein thrombosis (DVT) was the third most common cardiovascular disease globally 1 while the mortality rate can be higher than 15% among patients with pulmonary embolism and haemodynamic instability 2. Historically, pulmonary embolism was managed primarily as an inpatient condition 3. In the past two decades, advancement in diagnostic modalities, risk stratification tools and introduction of new anticoagulants enabled a shift towards more individualised management approach for suspected pulmonary embolism 5. This review summarises strategies from various guidelines for managing suspected pulmonary embolism cases.
Introduction and Background
Pulmonary embolism is the third most common cardiovascular disease globally just behind myocardial infarction and stroke 1. The clinical presentation of pulmonary embolism could be variable, from asymptomatic or subtle presentation to severe life-threatening presentation such as obstructive shock and sudden cardiac arrest.
Given the subtle and nonspecific clinical presentations, it is important to have high suspicion of pulmonary embolism as a differential diagnosis for the patients with risk profiles for venous thromboembolism. Since risk of mortality can be high with pulmonary embolism with haemodynamic instability,providing prompt and effective treatment is essential.
Clinical presentations and Diagnostic work-up
The most common presentations for pulmonary embolism are dyspnoea, pleuritic chest pain, and haemoptysis followed by cough, syncope, unexplained hypoxia or angina like chest discomfort or cardiac arrest.
Depending on the clinical stability, two different approaches could be considered for management of suspected PE cases.
In general, when the patients are haemodynamically unstable, the mainstay of treatment would be resuscitative measures and urgent thrombolysis. Bedside echocardiogram and CTPA, if it can be urgently organised, can be useful.
When the patients are haemodynamically stable, systemic approach integrating clinical assessment with clinical probability scoring should be considered to determine the diagnostic pathway. If there is high clinical probability of PE, definitive imaging like CTPA or VQ should be done without need for checking D-dimer. If the probability is low or intermediate, D-dimer can be checked first. In such cases, negative D-dimer can effectively rule out PE without need for further imaging. Depending on clinical judgement and risk of deterioration in suspected PE patients, either outpatient or inpatient management pathways can be considered. Urgent thrombolysis is not recommended in normotensive patients. Parenteral or oral anticoagulant agents are the first line of therapy in this group.
Details of the diagnostic measures and therapeutic options are discussed as below.
Haemodynamically unstable patients
The patients with suspected pulmonary embolism are considered to be haemodynamically unstable if they present with either of the following conditions [1]:
(1) Cardiac arrest: requiring cardiopulmonary resuscitation
(2) Obstructive shock: Systolic BP < 90 mmHg or vasopressors required to achieve a BP ≥90 mmHg despite adequate filling status AND End-organ hypoperfusion (altered mental status; cold, clammy skin; oliguria/anuria; increased serum lactate)
(3) Persistent hypotension: Systolic BP < 90 mmHg or systolic BP drop ≥40 mmHg, lasting longer than 15 min and not caused by new-onset arrhythmia, hypovolaemia, or sepsis
For these patients, both diagnostic and therapeutic measures should take place simultaneously 5. If pulmonary embolism is the most likely cause for instability, the initial approach should be emphasized on the restoration of perfusion alongside the resuscitative measures. In case of cardiac arrest, the cardiopulmonary resuscitation effort should continue for 60-90 minutes after administering thrombolytic measures.
In terms of diagnostic measures, definitive diagnostic imaging, usually CT pulmonary angiogram (CTPA), should be organised urgently. When emergency CTPA is not feasible, bedside echocardiogram assessing RV dysfunction and overload can be helpful to justify empirical thrombolysis or reperfusion therapy in such high-risk patients.1 The absence of evidence of RV dysfunction or overload on echo practically excludes the PE as the cause of haemodynamic instability 6 in this cohort of patients.
Haemodynamically stable patients
On the other hand, in this group of haemodynamically stablepatients, systematic approach to reach the definitive diagnosis is recommended 1,5.
Once pulmonary embolism is suspected, an assessment of general medical history, physical examination and, when necessary, investigations such as chest X-ray and ECG, must be carried out.
Integration of such clinical evaluation with validated probability scoring system can guide the choice of further investigations for diagnosis. Here, we will briefly discuss about some of the commonly used validated probability scoring systems.
Pulmonary embolism rule-out criteria (PERC)
If the clinical probability of pulmonary embolism is low on gestalt suspicion or on a clinical prediction rule such as Well’s score, PERC rule can be applied to decide whether further investigations are justified 7. If there are none of the eight-items criteria of PERC met, the diagnosis of pulmonary embolism can be ruled out without the need to check D-dimer and the metanalysis showed that this approach had only a failure rate 0.7% 8.
Limitation: It was only validated for the use in clinical settings where the incidence of PE is relatively low, such asthe emergency department 7,8. It is not advisable to use this to rule out PE among high-risk groups like inpatients with suspected pulmonary embolism or pregnant women.8
Wells score, Simplified Wells score and Revised Geneva scorefor pulmonary embolism These clinical decision rules help differentiating the pre-testclinical probability of pulmonary embolism, taking account ofclinical features and risk factors for pulmonary embolism. Wells score and Simplified Wells score emphasize on sevenkey features while modified Geneva score uses nine variables.Modified Geneva score has been shown to be as effective as the Wells score and is less reliant on the experience of the clinician assessing the patient 9. Depending on the total scores, they classified the suspected pulmonary embolism cases into different probability groups.
Wells score – total score of <2 “low probability”; 2-6“intermediate probability”, and >6 “high probability”.
Modified/Simplified Wells – total score <4 “PE unlikely” and,4 or above “PE likely” Revised Geneva score- total score 0-3 “low probability”, 4-10 “intermediate probability” and 11 and above “high probability”
If the integrated clinical assessment with the scoring system suggests low or intermediate clinical probability of PE, it is recommended to check D-dimer, preferably using high sensitivity D-dimer assay. If D-dimer level is below 500 ng/ml, PE is excluded, and alternative diagnoses are to be considered. If D-dimer is above 500 ng/ml, further definitive investigations like CTPA or VQ are recommended.
If there is high clinical probability of PE, direct proceeding to definitive investigations is recommended.
YEARS algorithm
In YEARS algorithm, differential cut-off values for D-dimer are used depending on whether one or more of the YEARS criteria are met.
Criteria include the clinical evidence of deep vein thrombosis (DVT), haemoptysis, whether the pulmonary embolism is the most likely diagnosis.
Pulmonary embolism is ruled out if there are no such criteria met and D-dimer value is less than 1000 ng/ml, or if there are one or more criteria met and D-dimer value is less than 500 ng/ml. Otherwise, further investigations like CTPA/VQ scan need to be done.
The initial validation study showed that this approach resulted the absolute 14% reduction of CTPA examinations in all ages with a low 3-month thromboembolic risk of 0.61% 10. The further external validation by Maddy et al. 11showed similar finding that the YEARS algorithm allowed safely ruling out PE with a low 3-month thromboembolism risk of 1.2%. Interestingly, this study showed that all of those who did not meet criteria for further investigations as per YEARS algorithm but had diagnosed with VTE at 3-month follow up had D-dimer level above the age-adjusted values. Thus, caution is needed when there are no YEARS items and D-dimer <1000 ng/ml, but if D-dimer is still above age-adjusted value.
Age-adjusted D-dimer
As D-dimer level increases with age, even in the absence of pathology, the use of fixed cut-off D-dimer value can increase the rate of false positive among elderly patients, leading to unnecessary investigation burdens. The evidence reviews by NICE (National Institute for Health and Care Excellence) showed that the age adjusted D-dimer can be considered in lieu of fixed cut off d-dimer level among people aged over 50 2. This approach has only marginal reduction of sensitivity (96% vs 98%) with better specificity. Given the lack of evidence, this age-adjusted D-dimer cannot be applied in people under the age of 50.
Age adjusted D-dimer for people over age of 50 = age (years) x 10 ng/ml
Specific population
Pregnancy
It is more gestalt suspicion rather than clinical decision rules in assessing probability of pulmonary embolism among pregnant people given the overlapping clinical features between physiological changes of pregnancy and pulmonary embolism. The use of D-dimer is also limited as D-dimer level increases with the gestational age, reaching to the peak in third trimester 12,13,14. This has led to proceeding to the CTPA or VQ scan in most pregnant people with suspected pulmonary embolism.
In recent years, the successful use of the revised Geneva score or modified YEARS criteria (clinical signs of deep venous thrombosis [DVT], haemoptysis, and PE as the most likely diagnosis) in conjunction with both D-dimer and targeted PE imaging has gained traction and may reduce the number of unnecessary scans performed for PE during pregnancy.
Meta-analysis of 45 studies showed that negative D-dimer in conjunction with low clinical suspicion can rule out pulmonary embolism with sensitivity 99.5% and NPV 100% while the 3-month VTE rate was only 0.32% 6. Otherwise, further investigations like Compression ultrasound or CTPA or VQ are recommended to rule in/out pulmonary embolism.
In terms of choice of imaging, European Society of cardiology (ESC) 2019 guidance recommended that both CTPA and VQ scan can be considered for pregnant people as the radiation risk to the mother or the fetus was not clinically significant.
It is the radiation dose above 50 milligray (mGy) which is required to cause the complication. In a meta-analysis by Tromeur et al. 15 the fetal radiation dose from CTPA ranged from 0.002 to 0.51 mGy and that of V/Q scintigraphy from 0.2 to 0.7 mGy. The mean maternal dose ranged from 0.23 to 9.7 mGy with CTPA and 0.9 to 5.8 mGy with V/Q scan. Hence in general, the risk of radiation (from V/Q scanning and CTPA) and iodinated contrast is considered low and must be weighed against the 20 to 30 percent risk of maternal mortality of untreated PE.
Choice of diagnostic imaging study
CTPA
CT pulmonary angiogram (CTPA) is the mainstay of diagnostic imaging study to confirm pulmonary embolism which appears as a filling defect on CTPA. It can assist not only with diagnostic confirmation but also with prognostication by providing information about right ventricular strain (RV:LV ratio, straightening of ventricular septum). It can also show other alternative causes or pathology to explain the presented symptoms even when pulmonary embolism is ruled out.
Improving technology with the CT scanner means less radiation exposure compared to the older scanners.
Caution with CTPA would be risk of allergic response to CT contrast and contrast-induced renal impairment. In case of severe renal impairment or allergic response to CT contrast, alternative imaging modalities such as V/Q scan need to be considered.
V/Q scan
Ventilation-perfusion (V/Q) scan is a nuclear scan measuring and comparing the distribution of blood flow and ventilation across the lung fields. Mismatch between the perfusion and ventilation suggests the presence of pulmonary embolism.
Its use is limited in case of pre-existing lung pathology or known pulmonary AV shunt or abnormal chest X-ray.
Royal College of Obstetrics and Gynaecology guideline in 2015 recommended that it is prudent to consider the V/Q scan as the investigation of first choice for young women, especially if there is a family history of breast cancer or if the patient has had a previous chest CT scan, in view of relatively lower radiation risk to breast tissue with V/Q scan.
European Society of cardiology guidelines in 2019 suggested both CTPA and V/Q scans are equally safe during pregnancy and highlighted that the V/Q scan can be inconclusive in some cases which would result in another alternative imaging such as CTPA.
Other imaging modalities
Chest X-ray (CXR)
CXR is useful to rule out other differential diagnosis like pneumothorax, pneumonia, or pulmonary oedema while it is not diagnostic for pulmonary embolism. However, there are some radiographic findings suggestive of pulmonary embolism. These include:
(1) Fleischner sign: An enlarged pulmonary artery to pulmonary hypertension or distension of the vessel by pulmonary embolus.
(2) Westermark sign: Regional oligemia from PE
(3) Hampton hump: a peripherally located wedge shaped opacity suggesting pulmonary infarction.
Magnetic resonance angiogram (MRA)
MRI allows for an accurate and reproducible assessment of the right and left ventricular myocardial function, hypertrophy, and fibrosis described as delayed enhancement.
However, in comparison with CTPA, MRPA had lower sensitivities for detecting PE, especially in peripheral pulmonary arteries.
Conventional pulmonary angiography
Due to the invasive nature of the conventional pulmonary angiogram, its use is limited these days for certain setting like when a concomitant endovascular treatment is planned.
Treatment algorithms
Haemodynamically unstable patients
Resuscitative measures and early reperfusion therapy are the main strategy. Systemic thrombolysis is the treatment of choice for reperfusion in most clinical settings. Tissue-type plasminogen activator such as Alteplase or Tenecteplase are preferred to first generation thrombolytic agents such as streptokinase or urokinase. Surgical pulmonary embolectomy or percutaneous catheter-directed thrombolysis are alternative reperfusion treatments in patients with contraindications to systemic thrombolysis if expertise with either of these methods and the appropriate resources are available on-site 1
Following reperfusion therapy and haemodynamic stabilisation, consider switching the treatment to oral anticoagulation therapy.
Haemodynamically stable patients
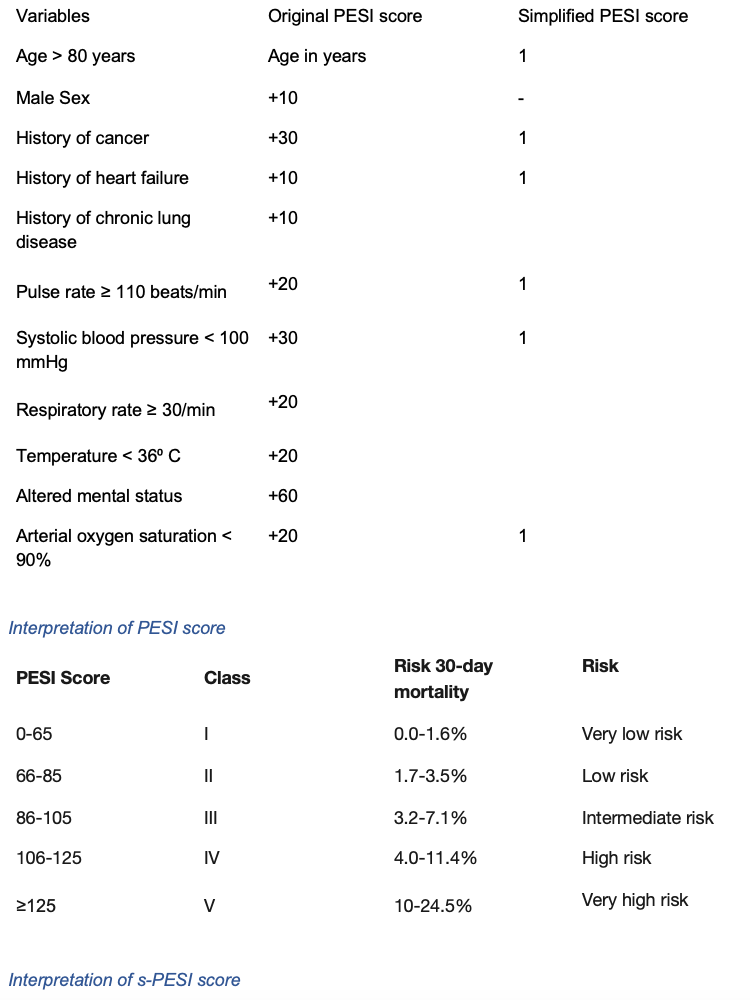
Clinical judgement in conjunction with use of scoring system for prognosis like Hestia criteria or pulmonary embolism severity index (PESI) or simplified-PESI (s-PESI) can help triaging the patients for either inpatient or outpatient management pathways. Patients with one of the high-risk factors for PE or aggravating factors or co-morbidity should be admitted to a hospital.Treatment options are pharmacological treatment, mechanical interventions, or thrombolytic therapy.
Pharmacological option
This includes:
- Parenteral anticoagulation: unfractionated heparin, low molecular weight heparin (LMWH), or fondaparinux
- Oral anticoagulation: vitamin K antagonist such aswarfarin, or direct oral anticoagulants (DOACs) such asrivaroxaban, apixaban, edoxaban, or dabigatran.
Among oral anticoagulants, rivaroxaban and apixaban do not need any loading with LMWH. For edoxaban or dabigatran, LMWH needs to be given for the first 5 days followed by edoxaban or dabigatran. In case of warfarin, LMWH needs to be given with warfarin for at least 5 days or until the international normalised ratio (INR) is at least 2 in two consecutive readings, followed by warfarin on its own.
For those with intermediate to high-risk PE should be on LMWH in the first 2-3 days before switching to an oral anticoagulant given that the risk of deterioration is highest in the first 2-3 days. 1
For patients with severe renal impairment or in those with triple positive antiphospholipid syndrome, there is noevidence for use of DOACs (direct oral anticoagulants) and the recommendation for long-term anticoagulation of choice would be vitamin antagonist such as warfarin.
For pregnant patients with PE, only unfractionated heparin or low molecular weight heparin are recommended.
For those at extreme body weights (< 50 kg or >120 kg), regular monitoring of therapeutic levels is recommended to ensure effective anticoagulation [2]
Mechanical Interventions
Mechanical interventions include inferior vena cava filters,but the use is restricted to prospective clinical studies unless anticoagulation is contraindicated, or PE has occurred during anticoagulation treatment [2]
Thrombolytic therapy
Thrombolytic therapy might be systemic thrombolysis, or in case of contraindication to systemic thrombolysis, catheter-directed thrombolysis or open pulmonary embolectomy. Open pulmonary embolectomy is not used commonly in modern day practice.
In haemodynamically stable patients, there is no sufficient evidence to recommend using urgent thrombolytic therapy. In case of deterioration, rescue thrombolytic therapy can be considered.
Duration of anticoagulation therapy
The duration of treatment depends on the underlying conditions precipitating pulmonary embolism.
For people with provoked PE, stopping anticoagulation therapy can be considered at 3 months (3-6 months for active cancer) provided that the provoking factor is no longer present, and the clinical course has been uncomplicated.
For people with unprovoked PE, continuation of anticoagulation beyond 3 months should be considered, on the balance between the person’s risk of VTE recurrence and their risk of bleeding.
What happens next?
After initial diagnosis of PE with no clear risk factors, the possibility of occult malignancies (4-9% incidence as per Nick Van Es et al).16 and haematological pathologies should be considered.The initial step would be comprehensive history taking,physical examination, and investigations such as full blood count, renal and liver functions including prothrombin time and activated partial thromboplastin time.
If there are clinical features suggesting malignancy, further investigations would be recommended to rule out occult malignancy.
Antiphospholipid antibodies should be tested before starting anticoagulants if there is planning to stop anticoagulation treatment in the future. They should be reassessed in 3-6 months after starting treatment.
For those with plan for extended anticoagulation, liver and renal functions and bleeding risk should be reassessed at regular intervals.
Follow-up and long-term sequel
The patency of pulmonary arterial bed is restored in the majority of patients within the first few months. The incidence of persistent pulmonary arteries by organised thrombus causing chronic thromboembolic pulmonary hypertension (CTEPH) is very low with the cumulative incidence of between 0.1% and 9.1% in the first 2 years. Despite this rare occurrence of CTEPH, 20-75% of patients with diagnosed PE would report that the health status is worse at 6-months follow up period 17,18
Hence ESC 2019 1 recommends that follow up should be offered at 3-6 months’ time for review of the symptoms.Appropriate care like exercise rehabilitation, treatment of comorbidity, behavioral education, and modification of risk factor should be offered to patients with persisting dyspnoea or functional limitation and further diagnostic workup should be considered for early detection of CTEPH.
Echocardiogram can be used to estimate the probability of pulmonary hypertension. If there is high echo probability of pulmonary hypertension, Planar V/Q lung scan is a suitable first-line imaging modality for CTEPH as it has 96–97% sensitivity and 90–95% specificity for the diagnosis. Right heart catheterisation is the gold standard diagnostic test to confirm the diagnosis of CTEPH.
In confirmed CTEPH, surgical or medical measures with management of symptoms should be considered, preferably at a specialised centre of pulmonary hypertension.
References
Khin Pyae Sone1, Inzali Moe Moe Kyaw2
1. Stavros V Konstantinides, Guy Meyer, Cecilia Becattini, Héctor Bueno, Geert-Jan Geersing, Veli-Pekka Harjola, Menno V Huisman, Marc Humbert, Catriona Sian Jennings, David Jiménez, Nils Kucher, Irene Marthe Lang, Mareike Lankeit, Roberto Lorusso, Lucia Mazzolai, Nicolas Meneveau, Fionnuala Ní Áinle, Paolo Prandoni, Piotr Pruszczyk, Marc Righini, Adam Torbicki, Eric Van Belle, José Luis Zamoran, ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). European Heart Journal. 2020; 41, (4):543-603. https://doi.org/10.1093/eurheartj/ehz405
2. National Institute for Health and Care Excellence. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing [Internet]. London, NICE, 2020 [cited 2024 Sept 10]. [NG158]. Available from: https://www.nice.org.uk/guidance/ng158
3. McFadden PM, Ochsner JL. A history of the diagnosis and treatment of venous thrombosis and pulmonary embolism. Ochsner J. 2002 Winter; 4(1):9-13. Available from: McFadden PM, Ochsner JL. A history of the diagnosis and treatment of venous thrombosis and pulmonary embolism. Ochsner J. 2002 Winter;4(1):9-13. Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3399235/ (Accessed on September 20, 2024)
4. Roy PM, Moumneh T, Penaloza A, Sanchez O. Outpatient management of pulmonary embolism. Thromb Res. 2017; 155:92-100. doi: 10.1016/j.thromres.2017.05.001. Epub 2017 May 4. PMID: 28525830.
5. B Taylor Thompson, Christopher Kabrhel, Constantino Pena. Clinical presentation, evaluation, and diagnosis of the nonpregnant adult with suspected acute pulmonary embolism. In: Connor RF, editors. UpToDate [internet]. Waltham (MA): UpToDate Inc; 2024 (Accessed on September 13, 2024.)
6. Atul Malhotra, Steven E Weinberger. Pulmonary embolism in pregnancy: Clinical presentation and diagnosis. In: Jess Mandel, Charles J Lockwood, Nester L Muller, James D Douketis, Geraldine Finlay, Alana Chakrabarti, editors. UpToDate [internet]. Waltham (MA): UpToDate Inc; 2024 (Accessed on September 13, 2024)
7. Kline JA, Courtney DM, Kabrhel C, et al. Prospective multicenter evaluation of the pulmonary embolism rule-out criteria. J Thromb Haemost. 2008; 6(5): 772-780.Available from: https://www.jthjournal.org/article/S1538-7836(22)12632-2/fulltext (Accessed on September 20, 2024)
8. Perrine Truong, Lucia Mazzolai, Carme Font, et al. Safety of the pulmonary embolism rule-out criteria rule: Findings from the Registro Informatizado de la Enfermedad TromboEmbolica venosa (RIETE) registry.Academic Emergency medicine. 2023; 30(9): 935-945. Available from: https://doi.org/10.1111/acem.14744(Accessed on September 20, 2024)
9. Tak T, Karturi S, Sharma U, Eckstein L, Poterucha JT, Sandoval Y. Acute Pulmonary Embolism: Contemporary Approach to Diagnosis, Risk-Stratification, and Management. Int J Angiol. 2019;28(2):100-111. doi: 10.1055/s-0039-1692636. Epub 2019 Jul 5. PMID: 31384107; PMCID: PMC6679967.
10. van der Hulle T, Cheung WY, Kooij S, Beenen LFM, van Bemmel T, van Es J, Faber LM, Hazelaar GM, Heringhaus C, Hofstee H, Hovens MMC, KaasjagerKAH, van Klink RCJ, Kruip MJHA, Loeffen RF, Mairuhu ATA, Middeldorp S, Nijkeuter M, van der Pol LM, Schol-Gelok S, Ten Wolde M, Klok FA, Huisman MV; YEARS study group. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390(10091):289-297. doi: 10.1016/S0140-6736(17)30885-1.
11. Bellesini M, Verschuren F, Moumneh T, Meyer G, Righini M, Le Gal G. External validation of the YEARS diagnostic algorithm for suspected pulmonary embolism. J Thromb Haemost. 2020;18(12):3289-3295. doi: 10.1111/jth.15083.
12. Murphy N, Broadhurst DI, Khashan AS, Gilligan O, Kenny LC, O’Donoghue K. Gestation-specific D-dimer reference ranges: a cross-sectional study. BJOG. 2015;122(3):395-400. doi: 10.1111/1471-0528.12855.
13. Jeremiah ZA, Adias TC, Opiah M, George SP, MgbereO, Essien EJ. Elevation in D-dimer concentrations is positively correlated with gestation in normal uncomplicated pregnancy. Int J Womens Health. 2012;4(437-443). doi: 10.2147/IJWH.S32655.
14. Mohmad Sallih N, Subbiah I, Ali A, Jackson N. Reference ranges for D-dimer levels in Malaysian women in the three trimesters of pregnancy. Malays J Pathol. 2019;41(1):7-13.
15. Tromeur C, van der Pol LM, Le Roux PY, Ende-VerhaarY, Salaun PY, Leroyer C, Couturaud F, Kroft LJM, Huisman MV, Klok FA. Computed tomography pulmonary angiography versus ventilation-perfusion lung scanning for diagnosing pulmonary embolism during pregnancy: a systematic review and meta-analysis. Haematologica. 2019;104(1):176-188. 10.3324/haematol.2018.196121
16. Van Es N, Le Gal G, Otten HM, Robin P, Piccioli A, Lécumberri R, Jara-Palomares L, Religa P, Rieu V, Rondina MT, Beckers MM, Prandoni P, Salaun PY, Nisio MD, Bossuyt PM, Büller HR, Carrier M. Screening for cancer in patients with unprovoked venous thromboembolism: protocol for a systematic review and individual patient data meta-analysis. BMJ Open. 2017;7(6):e015562. doi: 10.1136/bmjopen-2016-015562. Erratum in: BMJ Open. 2017 Aug 11;7(8):e015562corr1. doi: 10.1136/bmjopen-2016-015562corr1. PMID: 28601834; PMCID: PMC5663010.
17. Klok FA, van Kralingen KW, van Dijk AP, HeyningFH, Vliegen HW, Huisman MV. Prevalence and potential determinants of exertional dyspnea after acute pulmonary embolism. Respir Med. 2010 Nov;104(11):1744-9. doi: 10.1016/j.rmed.2010.06.006. PMID: 20599368.
18. Kline JA, Steuerwald MT, Marchick MR, Hernandez-Nino J, Rose GA. Prospective evaluation of right ventricular function and functional status 6 months after acute submassive pulmonary embolism: frequency of persistent or subsequent elevation in estimated pulmonary artery pressure. Chest. 2009 Nov;136(5):1202-1210. doi: 10.1378/chest.08-2988. Epub 2009 Jun 19. PMID: 19542256; PMCID: PMC2818852.
Appendix A: Clinical Probability Scoring systems
PERC rule criteria
1. age >50 years
2. pulse >100 bpm
3. oxygen saturation <94% on room air
4. presence of unilateral leg swelling
5. presence of haemoptysis
6. history of recent trauma or surgery
7. Prior history of venous thromboembolism
8. History of exogenous oestrogen use
Wells scoring system


0-3 points – low pretest probability
4-10 points – intermediate pretest probability
≥ 11 points – high pretest probability
Appendix B: Prognostic/Severity Index Scoring Systems
Hestia criteria
If a patient with suspected pulmonary embolism does not meet any of Hestia criteria, the risk is low and outpatient management of PE can be considered.
1. Hemodynamically unstable
a. SBP <100 mmHg and HR >100
b. needing ICU care, or
c. clinician judgment
2. Thrombolysis or embolectomy needed (for reasons other than hemodynamic instability)
3. Active bleeding or high risk for bleeding
a. GI bleeding or surgery ≤2 weeks ago,
b. stroke ≤1 month ago,
c. bleeding disorder or platelet count <75 × 10⁹/L d. uncontrolled HTN (SBP >180 or DBP >110), or
e. clinician judgment
4. >24 hrs on supplemental oxygen required to maintain SaO₂ >90%
5. PE diagnosed while on anticoagulation
6. Severe pain needing IV pain medication required >24 hr
7. Medical or social reason for admission >24 hr (infection, malignancy, no support system)
8. Creatinine clearance <30 mL/min by Cockcroft-Gault
9. Severe liver impairment
10. Pregnant
11. Documented history of heparin-induced thrombocytopenia (HIT)
Pulmonary embolism severity index (PESI) and simplified PESI
If it is suggestive of very low or low risk, outpatient management of pulmonary embolism can be considered. This is also validated to predict the 30-day mortality risk.

Author Information
Khin Pyae Sone1, Inzali Moe Moe Kyaw2
1. IMT2, West Suffolk Hospital NHS trust
2.Consultant in Acute Medicine, West Suffolk Hospital NHS trust




