COVID 19 Vaccines: Where Are We Now?

Introduction
The pandemic of COVID 19 started from Wuhan Province of China in the latter part of December 2019. It spread to the different parts of the world. Globally, as of 10:43am CET, 24 November 2020, there have been 58,712,326 confirmed cases of COVID-19, including 1,388,528 deaths, reported to WHO.1 The Chinese scientists had shared the genetic sequence of SAR-COV2 virus since 12.1.2020, within two weeks after the announcement of clusters of cases of unusual pneumonia in Wuhan2. Since the availability of genetic sequence, many scientists all over the world have been trying to start developing diagnostic tests as well as steps for development of vaccines.
Status of Candidate Vaccines at 12th November
The WHO published a draft landscape of Covid 19 vaccines on 12 November 2020. According to this report, out of 200 candidate vaccines, 48 candidate vaccines are clinical evaluation and 164 candidate vaccines in pre-clinical evaluation. Eleven are in the Phase 3 trials – Sinovac, Wuhan Institute of Biological Products, Sinopharm, Beijing Institute of Biological Products, Sinopharm, Bharat Biotech, University of Oxford/AstraZeneca, CanSino Biological Inc. /Beijing Institute of Biotechnology, GamaleyaResearch Institute, Janssen Pharmaceutical Company, Novavax, Moderna/NIAID, BioNTech/FosunPharma/Pfizer. Out of the eleven vaccines in Phase 3 trials, four are from China, one from Russia, one from India, and four from the USA. Among those, four are inactivated vaccine – Sinovac, Sinopharm (Beijing), Sinopharm (Wuhan), Bharat Biotech, four vaccines using non-replicating viral vector Adenovirus- CanSinp, Oxford, GAMALEYA, Jansen. Novavxuse full length recombinant SARS CoV-2 glycoprotein nanoparticle vaccine adjuvanted with Matrix M. Moderna and Pfizer vaccines use m RNA.3
Steps in vaccine Developments
The general stages of the development cycle of a vaccine are as follows: Exploratory stage, Pre-clinical stage, Clinical development, Regulatory review and approval, Manufacturing and Quality control.
Clinical development is a three-phase process. During Phase I, small groups of people receive the trial vaccine. In Phase II, the clinical study is expanded and vaccine is given to people who have certain characteristics (such as older age and impaired physical health) similar to those for whom the new vaccine is intended. In Phase III, the vaccine is given to thousands of people to tested efficacy and safety.4 Many vaccines undergo Phase IV formal, ongoing studies after the vaccine is approved and licensed.4
Usually the whole process will take between five to ten years, but because of the urgency to produce an effective and safe vaccine, the whole process will be shortened and many manufacturers will seek emergency utilization licensure.5
ACT – accelerator and COVAX facility
Effective vaccines against COVID-19 are urgently needed to protect populations and restart economies. Where they can, governments are funding the development of candidate vaccines and making bilateral agreements with vaccine manufacturers. Research suggests that historically, vaccine programmes that have not yet entered human trials have a 7% probability of succeeding, which rises to 17% once they enter human trials.6
On 24 April, the World Health Organization (WHO) hosted the launch of the Access to COVID-19 Tools (ACT) Accelerator, a new global collaboration to accelerate development, production, and equitable access to COVID-19 diagnostics, therapeutics, and vaccines. Within the Accelerator, GAVI, CEPI, and WHO together with multinational and developing country vaccine manufacturers are working on the COVAX (Vaccine) Pillar to develop the COVAX Facility. The Facility provides governments with the opportunity to benefit from a large portfolio of COVID-19 candidate vaccines using a range of technology platforms, produced by more manufacturers across the world, with a bigger market to provide security of demand – often with greater scope than governments or regional groups can support on their own.6
The goal of the COVAX Facility is to ensure equitable access to all economies and ensure that income is not a barrier to access. The COVAX Facility serves as an invaluable insurance policy for participants to secure access to safe and effective COVID-19 vaccines through its actively managed portfolio of vaccine candidates across a broad range of technologies. The Facility provides all participants’ access to the same vaccine candidates, on the same accelerated timeline in an effort to end the COVID-19 pandemic as quickly as possible.
The goal of COVAX is to deliver two billion doses of safe, effective vaccines by end 2021, which have passed regulatory approval and WHO prequalification.6
Myanmar and COVAX Facility
As a low-middle income country Myanmar is eligible to get COVID vaccines through the COVAX facility with the minimal contribution from the country up to 3% of population during second quarter of the 2021 and another 17 % of population by the end of 2021. In addition to that the country can apply for additional doses with full contribution through pool procurement mechanism, through which we will get much cheaper price than bilateral procurement. Myanmar is now preparing the vaccines deployment plan for most effective ways of limited supply of COVID vaccine to COVID Facility.7
Prioritization of target population
The general guidance of SAGE for prioritization of target population is as follow.8

What are we doing now?
The COVID 19 second wave affected Myanmar in early September and confirmed cases have reached 82,236 and total deaths have reached 1784. (24.11.2020, 8 pm, MOHS). As Myanmar is a low-middle-income country, we are eligible for COVAX facility. In preparation for the possible purchase and distribution of COVID vaccines as early as possible, Myanmar is employing different strategies. The Covax National Co-ordinating Committee –CNCC, led by HE Minister of Health and Sports and COVAX National Technical Working Group (CTWG), led by Director General of Public Health and Medical Care, MOHS was formed on 9th October, 2020. The National Immunization Technical Advisory Group (NITAG), composed of experienced public health specialists, Pediatricians, Physicians, Microbiologists, Pathologists and Researchers, have been continuously monitoring the progress of various Covid vaccine developments and giving necessary advice to the CNCC. The Central EPI is working as the secretary to above committees and provides updated information and implementing the decisions of CNCC. Myanmar had already submitted our readiness assessment forms to World Bank and Asia Development Bank and preparation to submit detail vaccine deployment plan to COVAX facility.9
Current Status of Individual vaccines in the latest development
Current status of Vaccines in Phase 3 clinical trials are summarized in following tables 10
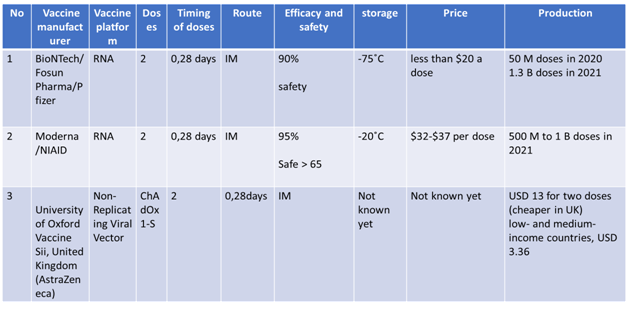
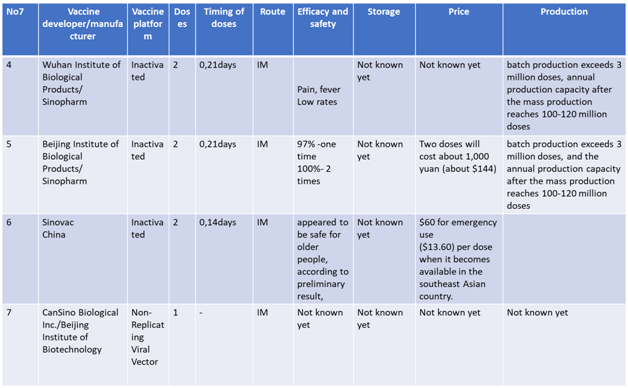
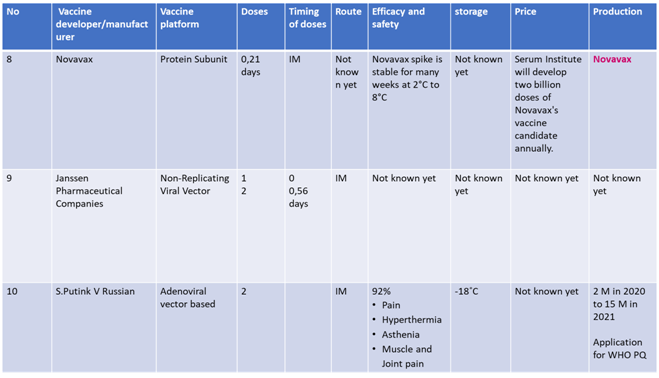
Advantages and Disadvantages of Various Vaccines
None of the vaccines at the present moment have completed the phase 3 trials. However, with the urgent requirement to control the pandemics, the foremost vaccines have applied for Emergency use listing and plan to roll out in the coming few weeks.5 Some are already used in parts of their own population and some part of the world after applying for EUL in local regulatory authorities. The m RNA based vaccines which have the highest technological innovation are likely to be safest, but as they require to be stored in -70 C(Pfizer), -20C (Moderna), which will be a disadvantage for lower income countries where ultra-low cold chain facilities are lacking. In addition, some of the manufacturers are not enrolled in the COVAX facility. Oxford/ Astra Zeneca, an adenovirus vector based vaccine, has an advantage of requiring usual cold chain system. It is already enrolled to the COVAX facility and likely to be cheaper in cost. The efficacy of this vaccine according to interim analysis is 70% to 90 % lower than Pfizer, Moderna and Sinovac which claimed to have more than 90 % efficacy. As the duration of vaccines development process is less than one year, there is no data on long term efficacy of any vaccine at the present moment.11-14
Conclusion
The burden of COVID-19 across the world is enormous and it is crucial to end this pandemic as soon as possible. Development of safe and effective COVID-19 vaccine will play pivotal role in tackling this pandemic. Our country, as a low middle income country with limited resources is trying to control this COVID epidemic with collective efforts of government and public. Although there are definite hope of availability of safe and effective COVID vaccine in coming few months there is still many uncertainties in coverage and timeline. The public needs to strictly follow the already proven infection control measures, even after COVID vaccines are widely available.
References
- World Health Organization.WHO Coronavirus Disease (COVID-19) Dashboard [Online]. Available from: https://covid19.who.int/?gclid=CjwKCAiA5IL-BRAzEiwA0lc WYszP_2QDRpKpSIERXLlvXo4nPtzvxbUNhAEyP5kBC2_zhWP120cryhoCYjEQAvD_BwE [Accessed 24 November 2020]
- World Health Organization. Timeline: WHO’s COVID-19 response? [Online]. Available from:https://www.who.int/emergencies/diseases/novel-coronavirus-2019/interactive-timeline [Accessed 26 November 2020]
- World Health Organization. Draft landscape of COVID-19 candidate vaccines. [Online]. Available from: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines [Accessed 26 November 2020].
- Centers for disease control and prevention. Vaccines and Immunization. [Online]. Available from: https://www.cdc.gov/vaccines/basics/test-approve.html [Accessed 28 November 2020].
- World Health Organization. Emergency Use Listing Procedure for vaccines. [Online]. Available from: https://www.who.int/teams/regulation-prequalification/ eul/eul-vaccines [Accessed 28 November2020].
- World Health Organization. The COVAX facility – World Health Organization. [Online]. Available from: https://www.who.int/docs/default-source/ coronaviruse/act-accelerator/covax-facility-background.pdf?sfvrsn=810d3c22_2 [Accessed 28 November 2020].
- GAVI the vaccine alliance. 92 low- and middle-income economies eligible to get access to COVID-19 vaccines through GAVI COVAX AMC. [Online]. Available from: https://www. gavi.org/news/media-room/92-low-middle-income-economies-eligible-access-covid-19-vaccines-gavi-covax-amc [Accessed 28 November 2020]
- World Health Organization. WHO SAGE ROADMAP FOR PRIORITIZING USES OF COVID-19 VACCINES IN THE CONTEXT OF LIMITED SUPPLY. (1st ed.). [Online]: 2020. Available from: https://www.who.int/docs/default-source/immunization/sage/covid/ sage-prioritization-roadmap-covid19 vaccines.pdf?Status= Temp&sfvrsn= bf227443_ 2 [Accessed 28 November 2020].
- Myanmar digital news. MoHS discusses preparations for COVID-19 vaccination plan. [Online]. Available from: https://www.mdn.gov.mm/en/mohs-discusses-preparations-covid-19-vaccination-plan [Accessed 28 November 2020].
- National Coordinating committee. (2020). ‘10 candidiates in phase III’. In Minutes of meeting on COVID vaccine introduction (1/2020) 25 November 2020. Ministry of Health and Sports, Myanmar.
- Pfizer. PFIZER AND BIONTECH ANNOUNCE VACCINE CANDIDATE AGAINST COVID-19 ACHIEVED SUCCESS IN FIRST INTERIM ANALYSIS FROM PHASE 3 STUDY. [Online]. Available from: hhttps://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-announce-vaccine-candidate-against [Accessed 26 November 2020]
- National Institute of Health. Promising Interim Results from Clinical Trial of NIH-Moderna COVID-19 Vaccine. [Online]. Available from: https://www.nih.gov/news-events/news-releases/promising-interim-results-clinical-trial-nih-moderna-covid-19-vaccine [Accessed 26 November 2020
- University of Oxford. Oxford University breakthrough on global COVID-19 vaccine. [Online]. Available from: https://www.ox.ac.uk/news/2020-11-23-oxford-university- breakthrough-global-covid-19-vaccine?hp [Accessed 26 November 2020]
- Palacios R, et al. Double-Blind, Randomized, Placebo-Controlled Phase III Clinical Trial to Evaluate the Efficacy and Safety of treating Healthcare Professionals with the Adsorbed COVID-19 (Inactivated) Vaccine Manufactured by Sinovac – PROFISCOV: A structured summary of a study protocol for a randomized controlled trial. Trials. 2020 Oct 15;21(1):853. doi: 10.1186/s13063-020-04775-4
Saw Win. MBBS, DCH, MMedSc(Paed.), MRCPCH, FRCP. Former Professor/Head, Department Paediatrics, University of Medicine
Member, Immunization Technical Advisory Group, WHO-SEAR
Vice Chairman, National Immunization, Technical Advisory Group (NITAG)
Aung Khin Thein. MBBS, MMedSc(Paed.), FRCPCH. Associate Professor, Department of Paediatrics, University of Medicine 2, Yangon
Kyi San Thi. MBBS, MMedSc(Paed.), MRCPCH. Assistant Lecturer, Department of Paediatrics, University of Medicine 2, Yangon

